- Home
- Editorial
- News
- Practice Guidelines
- Anesthesiology Guidelines
- Cancer Guidelines
- Cardiac Sciences Guidelines
- Critical Care Guidelines
- Dentistry Guidelines
- Dermatology Guidelines
- Diabetes and Endo Guidelines
- Diagnostics Guidelines
- ENT Guidelines
- Featured Practice Guidelines
- Gastroenterology Guidelines
- Geriatrics Guidelines
- Medicine Guidelines
- Nephrology Guidelines
- Neurosciences Guidelines
- Obs and Gynae Guidelines
- Ophthalmology Guidelines
- Orthopaedics Guidelines
- Paediatrics Guidelines
- Psychiatry Guidelines
- Pulmonology Guidelines
- Radiology Guidelines
- Surgery Guidelines
- Urology Guidelines
Fluciclovine PET/CT, for better management of prostate cancer : LOCATE Trial
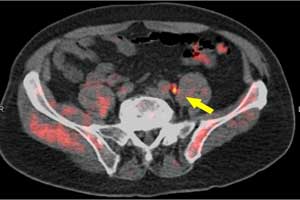
A new study presented at the 2018 Annual Meeting of the Society of Nuclear Medicine and Molecular Imaging (SNMMI) provides an insight for better imaging and treatment of the prostate cancer patients.
Data from the LOCATE trial demonstrates that the addition of fluorine-18 (18F)-fluciclovine positron emission tomography/computed tomography (PET/CT) to the diagnostic workup of patients with biochemical recurrence of prostate cancer locates previously undetected lesions and alter treatment management for the majority of patients.
The findings are also published in The Journal of Nuclear Medicine.
Austin R. Pantel, University of Pennsylvania, Philadelphia, U.S., and colleagues conducted the study to assess scan positivity rate and changes in management based on 18F-fluciclovine PET/CT.
Read Also: MRI guided biopsy improves detection rate in prostate cancer
"Recurrent prostate cancer poses an important medical challenge," explains Dr. Pantel. "Currently approved anatomical imaging procedures have limitations in identifying the sites of recurrence of prostate cancer after definitive treatment. This can make decision-making difficult when assessing these patients." He notes, "Up to 30 percent of patients with prostate cancer will develop local or distant recurrence within 10 years of radical prostatectomy or radiation therapy. Determining the location and extent of recurrent disease optimizes the selection of appropriate management for these men."
The LOCATE trial is a study of 18F-fluciclovine PET/CT in patients with biochemical recurrence (BCR) of prostate cancer after curative-intent primary therapy.
For the trial, 213 men with biochemically recurrent prostate cancer were evaluated with 18F-fluciclovine PET/CT after having negative or equivocal findings on conventional imaging, such as a bone scan, CT or magnetic resonance imaging (MRI).
Read Also: Breakthrough – New Imaging test developed for Prostate cancer
This first prospective multicenter study was conducted at 15 sites in the U.S., including both private practices and academic settings. It focused on the association between scan positivity and the clinical variables of recurrence site, practice setting, prostate-specific antigen (PSA) level, and Gleason score (system of grading prostate cancer). Questionnaires completed by treating physicians documented changes in management after the PET study.
Key Findings:
- 59 percent (126/213) of patients had their clinical management changed by findings from 18F-fluciclovine imaging. Of those changes, 78 percent (98/126) were classified as "major" (i.e., a change in treatment modality).
- The disease was detected in the prostate, as well as other tissue, including pelvic and abdominal lymph nodes and, less commonly, bone.
- Both positive and negative scans impacted patient management.
- No difference was seen in the rate of positive scans or in the rate of management changes between private practices and academic settings.
- No association was found between Gleason score at diagnosis and positive scans.
"While an investigation of the long-term clinical outcomes of these changes in management is warranted, these results indicate, for the first time in a prospective U.S. study, that decisions based on 18F-fluciclovine PET/CT imaging may facilitate more appropriate management in men with biochemical recurrence of their prostate cancer" adds Pantel. "The study also demonstrates the broad potential applicability of 18F-fluciclovine PET/CT imaging across a range of clinical settings."
He emphasizes, "Selecting appropriate treatment for men with recurrent prostate cancer is critical. Many options are available, and additional information, such as that provided by 18F-fluciclovine PET/CT, may help tailor personalized treatment plans. Clinical studies, such as the LOCATE trial, are important to investigate the role of the current generation of molecular imaging agents in guiding management for men with recurrent prostate cancer. This study highlights the key role of nuclear medicine in directing effective patient management and care."
Based on the study, the authors conclude that 18F-Fluciclovine PET/CT has proved clinically valuable in the detection of disease in patients with BCR of prostate cancer. The disease was detected in the prostate/bed, as well as in extraprostatic tissue, including pelvic and abdominal lymph nodes and, less commonly, in bone. There was a trend in PSA level and the rate of scan positivity. These findings may help guide the clinical application of 18F-fluciclovine PET/CT in its currently approved indication for imaging of men with BCR of prostate cancer.
For further reference follow the link: http://jnm.snmjournals.org/content/59/supplement_1/457.abstract?sid=268e6854-5a3a-48bc-ba68-f88a1016db4f

Disclaimer: This site is primarily intended for healthcare professionals. Any content/information on this website does not replace the advice of medical and/or health professionals and should not be construed as medical/diagnostic advice/endorsement or prescription. Use of this site is subject to our terms of use, privacy policy, advertisement policy. © 2020 Minerva Medical Treatment Pvt Ltd