- Home
- Editorial
- News
- Practice Guidelines
- Anesthesiology Guidelines
- Cancer Guidelines
- Cardiac Sciences Guidelines
- Critical Care Guidelines
- Dentistry Guidelines
- Dermatology Guidelines
- Diabetes and Endo Guidelines
- Diagnostics Guidelines
- ENT Guidelines
- Featured Practice Guidelines
- Gastroenterology Guidelines
- Geriatrics Guidelines
- Medicine Guidelines
- Nephrology Guidelines
- Neurosciences Guidelines
- Obs and Gynae Guidelines
- Ophthalmology Guidelines
- Orthopaedics Guidelines
- Paediatrics Guidelines
- Psychiatry Guidelines
- Pulmonology Guidelines
- Radiology Guidelines
- Surgery Guidelines
- Urology Guidelines
User friendly, Self-fitting hearing aid approved by FDA
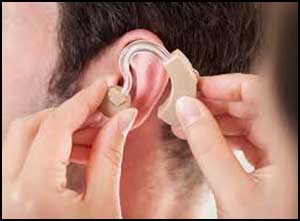
A new user-friendly hearing aid intended to amplify sounds for individuals 18 years or older with perceived mild to moderate hearing impairment has been allowed marketing by the U.S. Food and Drug Administration. This is the first hearing aid that enables users to fit, program and control the hearing aid on their own, without assistance from a health care provider.
The Bose Hearing Aid is a user-fitted wireless air conduction hearing aid that works by capturing sound vibrations through one or more microphones. The signal is processed, amplified, and played back through an earphone placed in the ear canal. Patients can adjust the hearing aid through a mobile application on their phone. This technology enables users to fit the hearing aid settings themselves, in real-time and in real-world environments without the assistance of a healthcare professional.
Hearing loss can be caused by aging, exposure to loud noises, certain medical conditions and other factors. Hearing loss may be permanent or temporary. Individuals with permanent hearing loss can use hearing aids to help them hear the speech and sounds around them better, enabling them to communicate more effectively with others.
“Hearing loss is a significant public health issue, especially as individuals age,” said Malvina Eydelman, M.D., director of the Division of Ophthalmic, and Ear, Nose, and Throat Devices at the FDA's Center for Devices and Radiological Health. “Today’s marketing authorization provides certain patients with access to a new hearing aid that provides them with direct control over the fit and functionality of the device. The FDA is committed to ensuring that individuals with hearing loss have options for taking an active role in their health care.”
While users may fit, program and control the new device on their own, it must comply with applicable federal and state laws regarding the sale of hearing aids, including state laws that might require hearing aids to be purchased from or dispensed by a licensed hearing aid dispenser.
In authorizing marketing of the Bose device, the FDA reviewed data from clinical studies of 125 patients, which demonstrated that outcomes with self-fitting of the Bose Hearing Aid are comparable on average to those with a professional fitting of the same device with respect to the amount of amplification selected, speech in noise testing and overall benefit. In addition, when participants self-fit the Bose Hearing Aid, they generally preferred those hearing aid settings over the professionally-selected settings. The Bose Hearing Aid has been labelled to inform the consumer when to consult a hearing healthcare professional.
The device was reviewed under the FDA’s De Novo premarket review pathway, a regulatory pathway for some low- to moderate-risk devices that are novel and for which there is no prior legally marketed device.
The FDA granted marketing authorization of the Bose Hearing Aid device to Bose Corporation.

Disclaimer: This site is primarily intended for healthcare professionals. Any content/information on this website does not replace the advice of medical and/or health professionals and should not be construed as medical/diagnostic advice/endorsement or prescription. Use of this site is subject to our terms of use, privacy policy, advertisement policy. © 2020 Minerva Medical Treatment Pvt Ltd