- Home
- Editorial
- News
- Practice Guidelines
- Anesthesiology Guidelines
- Cancer Guidelines
- Cardiac Sciences Guidelines
- Critical Care Guidelines
- Dentistry Guidelines
- Dermatology Guidelines
- Diabetes and Endo Guidelines
- Diagnostics Guidelines
- ENT Guidelines
- Featured Practice Guidelines
- Gastroenterology Guidelines
- Geriatrics Guidelines
- Medicine Guidelines
- Nephrology Guidelines
- Neurosciences Guidelines
- Obs and Gynae Guidelines
- Ophthalmology Guidelines
- Orthopaedics Guidelines
- Paediatrics Guidelines
- Psychiatry Guidelines
- Pulmonology Guidelines
- Radiology Guidelines
- Surgery Guidelines
- Urology Guidelines
Ultrasound-triggered liposomes- a novel technique of local anesthesia
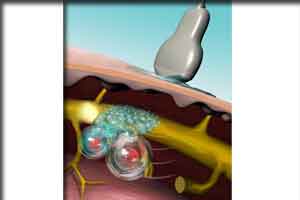
Researchers at Boston Children's Hospital have found a new way to non-invasively relieve pain at local sites in the body; such systems could one day improve pain management by replacing addictive opioids and short-lasting local anesthetics.
The novel system uses ultrasound to trigger the release of nerve-blocking agents injected into specific sites of the body ahead of time when and where pain relief is needed most. A paper describing the findings was published online today in Nature Biomedical Engineering.
"Opioid abuse is a growing problem in healthcare," says the paper's senior author, Daniel Kohane, MD, PhD, a senior associate in critical care medicine at Boston Children's and professor of anesthesiology at Harvard Medical School. "In the future, this system could potentially combat that by giving patients access to non-opioid, effective nerve-blocking drugs."
"One of the most interesting aspects about this system is that the degree of nerve block can be controlled just by adjusting the duration and intensity of the ultrasound," says the paper's co-first author, Alina Rwei, a graduate researcher in Kohane's lab.
Ultrasound is commercially available and widely used in various clinical and therapeutic settings, making it an attractive technology to use as a drug "trigger."
"We envision that patients could get an injection at the hospital and then bring home a small, portable ultrasound device for triggering the nerve-blocking agent," Rwei says. "This could allow patients to manage their pain relief at-will, non-invasively."
Tailoring a sono-sensitive drug delivery system
To create the ultrasound-triggered pain relief system, Kohane's team developed liposomes artificial sacs that are micrometers in size and filled them with a nerve-blocking drug. The walls of the liposomes contain small molecules called sono-sensitizers, which are sensitive to ultrasound.
"Once the drug-filled liposomes are injected, ultrasound can be applied to penetrate tissue and cause the sensitizers to create reactive oxygen species, which react with lipids in the walls of the liposomes," Kohane says. "This opens the surface of the liposomes and releases the nerve-blocking drug into the local tissue, reducing pain."
The small sono-sensitizer molecules that the team built into the liposomes are the active component of an already-FDA-approved drug that is currently used in photodynamic therapy. Right now, the pain treatment system developed by Kohane's team can be activated by ultrasound up to three days after injection of liposomes, making it well-positioned for future translation as a post-operative pain management strategy.
"Out of all the particle delivery systems, I think liposomes are one of the most clinically-acceptable and customizable options out there," Rwei says. "Our research indicates that liposomes can be tailored to respond to near-infrared light, ultrasound and even magnetic triggers."

Disclaimer: This site is primarily intended for healthcare professionals. Any content/information on this website does not replace the advice of medical and/or health professionals and should not be construed as medical/diagnostic advice/endorsement or prescription. Use of this site is subject to our terms of use, privacy policy, advertisement policy. © 2020 Minerva Medical Treatment Pvt Ltd