- Home
- Editorial
- News
- Practice Guidelines
- Anesthesiology Guidelines
- Cancer Guidelines
- Cardiac Sciences Guidelines
- Critical Care Guidelines
- Dentistry Guidelines
- Dermatology Guidelines
- Diabetes and Endo Guidelines
- Diagnostics Guidelines
- ENT Guidelines
- Featured Practice Guidelines
- Gastroenterology Guidelines
- Geriatrics Guidelines
- Medicine Guidelines
- Nephrology Guidelines
- Neurosciences Guidelines
- Obs and Gynae Guidelines
- Ophthalmology Guidelines
- Orthopaedics Guidelines
- Paediatrics Guidelines
- Psychiatry Guidelines
- Pulmonology Guidelines
- Radiology Guidelines
- Surgery Guidelines
- Urology Guidelines
Path-breaking STEMI Study paves way for better Heart Attack Care in India

New Delhi : India is a large country with a very high burden of non-communicable disease, particularly cardiovascular diseases. It is estimated that there are over 60 million patients with heart disease in India with over 2 million heart attacks every year. The low rates of healthcare spending by the state and national governments, uneven distribution of basic healthcare facilities, and the inability of large populations of rural and urban poor to access high-quality care makes the challenge of developing a viable heart attack program in India a daunting task.
Added to this is the disproportionately high prevalence of coronary artery disease in lower socioeconomic classes and consequent higher mortality due to poor access to affordable, high-quality health care. All these challenges toward acute treatment need to be balanced against additional health priorities toward prevention, and this scenario in India represents a prototypical case for many low- and middle-income countries.
Any heart attack treatment program in India should consider the huge manpower and Infrastructure deficiencies that exist in India. Blindly following the American or European system would not be feasible in this country. Furthermore, any heart attack management system developed in India could be used in other low- and middle-income countries that are confronted by similar magnitude of disease as well as financial and manpower constraints.
A landmark study published online on 8th March 2017 in the latest issue of the Journal of the American Medical Association (JAMA Cardiology) shows the results of a unique model of heart attack care run in the state of Tamil Nadu.
The year-long study, funded by the Indian Council of Medical Research (ICMR), was conducted by STEMI India, a not-for-profit organization set up by Dr. Thomas Alexander of KMCH Coimbatore and Dr. Ajit Mullasari of MMM Chennai. This study was done in four clusters in Tamilnadu and supported by the Government of Tamil Nadu through the Tamil Nadu Health Systems Project.
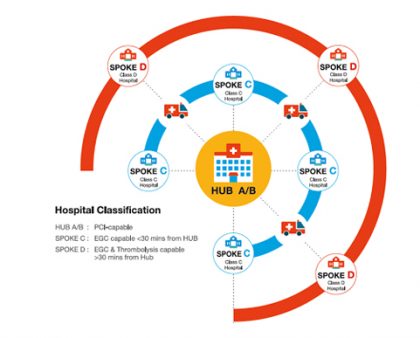
Traditionally, STEMI is treated by two strategies of re-perfusion. If a patient arrives at a hospital with a catheterisation laboratory (cath lab), Primary PCI is done. If a cath lab is not available, the patient is Thrombolysed and discharged. A third strategy, called the Pharmaco-Invasive method, involves Thrombolysis followed by PCI within 3-24 hours. The unique ‘STEMI India Model’ combines the two superior strategies of re-perfusion - Primary PCI and the Pharmaco-Invasive strategy - in a hub-and-spoke model. The Classic STEMI India model has a hub hospital, where a cath lab is available and primary PCI is done for patients directly presenting to these hospitals. These are linked to peripheral spoke hospitals, where thrombolysis is done and then shifted within 3-24 hours to the hub hospital for invasive treatment.
The 4 hub hospitals were Kovai Medical Center and Hospital Coimbatore, CMC Vellore, Madras Medical Mission Chennai and Government Stanley Medical College Chennai. These were connected to multiple peripheral hospitals, some Government and some private hospitals.
The three critical elements of the successful model were
- Utilisation of the unique ‘STEMI Kit’ for early diagnosis of STEMI at remote spokes by ECG’s transmitted to the hub and diagnosed by the ‘on call’ cardiologist located in the hub hospital. These “STEMI Kits” – a Multifunctional 12-lead ECG-integrated multi-parameter monitoring unit with data entry capability – are installed at the ‘STEMI hospitals’ and ambulances. The management algorithms and data entry, stored in the cloud, is also done using the same device.
- Utilisation of the BPL insurance to ensure that every patient can access the STEMI care.
- 108 Emergency ambulance services for pick up and transfer of patients. These ambulances had trained paramedics and were equipped with the ‘STEMI Kits’.
Data was collected from the 4 hub hospitals and 35 spoke hospitals, before implementation of the program and then after implementation of the heart attack program. The pre-implementation data collection was for an average period of 15 weeks and the post implementation period of 32 weeks. A total of 2,420 consecutive patients presenting with heart attack (898 pre- and 1,522 post-implementation phases, respectively) were enrolled between August 2012 and June 2014.
Analysis of the data showed that:
- It is possible to establish a system of care in India by combining the two strategies – Primary PCI and Pharmaco-invasive.
- Pharmaco-invasive and Primary PCI have similar Mortality and MACE events, both significantly better than those of standalone thrombolysis.
- Significantly greater number of patients utilised the superior strategies of Primary PCI or Pharmaco-invasive therapy as compared to stand-alone thrombolysis – 35% to 61%. This increased in the spoke hospitals from 3.5% to 31%.
- Significant numbers of the rural poor utilized the Below-Poverty-Line (BPL) insurance (0% to 60%) and could access the STEMI system of care and get best-in-class treatment of STEMI.
- The implementation of the STEMI India model of care produced an absolute mortality reduction of 3.4% (relative risk reduction of 19%) and an absolute MACE reduction of 4.3% (relative risk reduction of 21%).
One-year follow-up showed that, as compared to pre-implementation, the STEMI India model reduced patient death rates by 19%. This was because more patients, especially those presenting at peripheral spoke hospitals, received the superior treatment strategies of primary PCI or the pharmaco-invasive method, rather than stand-alone thrombolysis.
To read the full Model click on the following link
Publication:
A System of Care for Patients with ST-Elevation Myocardial Infarction in India: The Tamilnadu – ST-Elevation Myocardial Infarction Program
Thomas Alexander, Ajit S Mullasari, George Josep, Kumaresan Kannan4, Ganesh Veerasekar, Suma M Victor, Colby Ayers, Viji Samuel Thomson, Vijaykumar Subban, Justin Paul Gnanaraj, Jagat Narula, Dharam J Kumbhani, Brahmajee K Nallamothu for the STEMI-INDIA Investigators

Disclaimer: This site is primarily intended for healthcare professionals. Any content/information on this website does not replace the advice of medical and/or health professionals and should not be construed as medical/diagnostic advice/endorsement or prescription. Use of this site is subject to our terms of use, privacy policy, advertisement policy. © 2020 Minerva Medical Treatment Pvt Ltd