- Home
- Editorial
- News
- Practice Guidelines
- Anesthesiology Guidelines
- Cancer Guidelines
- Cardiac Sciences Guidelines
- Critical Care Guidelines
- Dentistry Guidelines
- Dermatology Guidelines
- Diabetes and Endo Guidelines
- Diagnostics Guidelines
- ENT Guidelines
- Featured Practice Guidelines
- Gastroenterology Guidelines
- Geriatrics Guidelines
- Medicine Guidelines
- Nephrology Guidelines
- Neurosciences Guidelines
- Obs and Gynae Guidelines
- Ophthalmology Guidelines
- Orthopaedics Guidelines
- Paediatrics Guidelines
- Psychiatry Guidelines
- Pulmonology Guidelines
- Radiology Guidelines
- Surgery Guidelines
- Urology Guidelines
New Dental material may keep Tooth alive after Root Canal
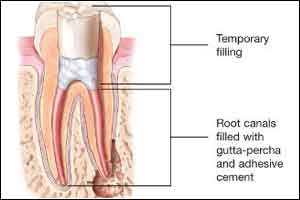
Scientists have reportedly developed a new biomaterial that may keep tooth alive after Root Canal. The new peptide hydrogel is designed to stimulate the growth of new blood vessels and dental pulp within a tooth after the procedure. A root canal is a dental procedure which is lengthy and sometimes painful surgery relieves the agony of an infection, but it results in a dead tooth with no living soft tissue, or dental pulp, inside.
The dentist drills off the top of an infected tooth to access the soft tissue inside during a root canal. They then remove the infected dental pulp and fills the space with tiny rubber rods called gutta percha and cap the repaired tooth with a crown.
“What you end up with after a root canal is a dead tooth,” Vivek Kumar, Ph.D., the project’s principal investigator, says. “It’s no longer responsive. There are no nerve endings or vascular supply. So the tooth is very susceptible to subsequent infection and, ultimately, falling out.”
The researchers have presented the results at the 256th National Meeting & Exposition of the American Chemical Society (ACS) featuring more than 10,000 presentations on a wide range of science topics.
A brand-new video on the research is available at http://bit.ly/acsrootcanal.
Kumar and Peter Nguyen at the New Jersey Institute of Technology have presented the work at the meeting. They wanted to develop a material that could be injected in place of the gutta-percha which would stimulate both new blood vessel growth, and or proliferation of dental pulp stem cells, within the tooth.
“We asked the question if we can stimulate angiogenesis in a limb, can we stimulate angiogenesis in other regions that have low blood flow?” Kumar says. “One of the regions we were really interested in was an organ in and of itself, the tooth.” So Kumar and Nguyen added another domain to the self-assembling angiogenic peptide: a piece of a protein that makes dental pulp stem cells proliferate.
When the team added the new peptide to cultured dental pulp stem cells, they found that the peptide not only caused the cells to proliferate but also activated them to deposit calcium phosphate crystals — the mineral that makes up tooth enamel. However, when injected under the skin of rats, the peptide degraded within one to three weeks. “This was shorter than we expected, so we went back and redesigned the peptide backbone so that we currently have a much more stable version,” says Kumar.
Now, the team is injecting the peptide hydrogel into the teeth of dogs that have undergone root canals to see if it can stimulate dental pulp regeneration in a living animal. If these studies go well, the researchers plan to move the hydrogel into human clinical studies. They have filed a patent for the redesigned peptide.
The hydrogel in its current form likely won’t reduce the invasiveness or pain of a root canal, but Kumar and Nguyen are planning future versions of the peptide that contain antimicrobial domains. “Instead of having to rip out everything inside the tooth, the dentist could go in with a smaller drill bit, remove a little bit of the pulp and inject our hydrogel,” Kumar says.
The antimicrobial portion of the peptide would kill the infection, preserving more of the existing dental pulp, while helping grow new tissue. And the root canal may no longer be such a dreaded procedure.
Kumar drew on his previous experience developing a hydrogel that stimulates angiogenesis when injected under the skin of rats and mice. The hydrogel, which is liquid during injection, contains peptides that self-assemble into a gel at the injection site. The peptides contain a snippet of a protein called vascular endothelial growth factor, that stimulated angiogenesis and persisted under the rodents’ skin for as long as three months.

Disclaimer: This site is primarily intended for healthcare professionals. Any content/information on this website does not replace the advice of medical and/or health professionals and should not be construed as medical/diagnostic advice/endorsement or prescription. Use of this site is subject to our terms of use, privacy policy, advertisement policy. © 2020 Minerva Medical Treatment Pvt Ltd