- Home
- Editorial
- News
- Practice Guidelines
- Anesthesiology Guidelines
- Cancer Guidelines
- Cardiac Sciences Guidelines
- Critical Care Guidelines
- Dentistry Guidelines
- Dermatology Guidelines
- Diabetes and Endo Guidelines
- Diagnostics Guidelines
- ENT Guidelines
- Featured Practice Guidelines
- Gastroenterology Guidelines
- Geriatrics Guidelines
- Medicine Guidelines
- Nephrology Guidelines
- Neurosciences Guidelines
- Obs and Gynae Guidelines
- Ophthalmology Guidelines
- Orthopaedics Guidelines
- Paediatrics Guidelines
- Psychiatry Guidelines
- Pulmonology Guidelines
- Radiology Guidelines
- Surgery Guidelines
- Urology Guidelines
Neonatal cholestasis - Standard Treatment Guidelines
Neonatal cholestatis is a pathological condition in the newborn where in bile flow from the liver is reduced. Neonatal Cholestasis Syndrome (NCS) includes a wide spectrum of clinical conditions ranging from congenital malformations of the hepatobiliary tree, infections, inborn errors of metabolism to some of the recently identified clinical conditions with or without genetic predilection. Most of these disorders have linkage with insults during antenatal, natal and postnatal periods. NCS has largely remained ignored in our country.
Ministry of Health and Family Welfare, Government of India has issued the Standard Treatment Guidelines for Neonatal cholestasis. Following are the major recommendations :
Case definition:
Neonatal cholestasis refers to conjugated hyperbilirubinemia >1.5 – 2 mg% and/or direct component of more than 20% of total bilirubin in a newborn/ infant with passage of high coloured urine with or without clay stools.
Incidence of The Condition In Our Country
NCS constitutes 30% of referrals with hepatobiliary disorders in India. The average age of presentation to a specialized center is 3.5 months (range birth to 15 months) with a consequent delay of 3 months in referral (medical and surgical centers). Based on consensus conference by paediatric gastroenterology, out of 1008 cases analysed in our country
- Hepatocellular causes: 53 %( neonatal hepatitis-47%, metabolic-4%, others-2%)
- Obstructive causes: 38 %( billiary atresia-34%, Choledochal cyst-4%)
- Ductal paucity: 3%
- Idiopathic: 6%
In neonatal hepatitis-
- Idiopathic giant cell hepatitis: 64%,
- TORCH: 22%
- Sepsis: 8%
- Others: 6%
Differential Diagnosis
A. Causes of Extra Hepatic Obstruction
- Biliary Atresia
- Choledochal Cyst
- Spontaneous perforation of bile ducts
- Biliary stenosis
- Inspissation of bile ducts
- Mass/peritoneal bands
B. Hepatocellular
1. Infective
- Sepsis
- TORCH
- Malaria
- UTI
- Hepatitis
- Other Viral infections
- HIV
2. Metabolic
- Galactossemia
- Hereditary Fructosemia
- Tyrosinemia
- Alfa 1 AT deficiency
- Bile acid disorders
- Fatty Acid Oxidation defects
- Cystic fibrosis
- Storage disorders
- Neonatal hemochromatosis
- Zelweger's disease
3. Miscellaneous
- TPN
- Shock
- Hypoperfusion
- Downs
- Congenital heart / valvular abn
4. Idiopathic
C. Paucity of Intrahepatic Ducts
- Syndromic - Alagille's syndrome, Byler's, Aagene's
- Non - Syndromic - a 1AT deficiency, Idiopathic, Familial
Prevention And Counseling
A high index of suspicion is necessary. Mothers must be informed about the need to seek medical attention if jaundice persists beyond two weeks of birth and / or baby passes pale stools and high coloured urine. If the previous sibling has had liver disease antenatal counselling and referral for further evaluation may be necessary.
Optimal Diagnostic Criteria, Investigations, Treatment & Referral Criteria
Diagnostic Criteria :
1. Clinical: Neonate with jaundice persistent beyond 2 weeks, dark colour urine
and/or pale stool
2. Screening Biochemistry: Serum bilirubin direct and indirect
Any child that meets with the clinical and /or biochemical criteria needs
investigation, treatment and referral.
Typical presentation:
- Newborn with jaundice/ high colored urine with or without clay colour stools beyond
two weeks of age. - Typically a child with biliary atresia is usually a term baby with normal weight, accepting
feeds well. - Pigmented stools do not rule out Biliary atresia, upto 30% of biliary atresia stools are
yellow in the early weeks.
Clinical examinations:
Clinical evaluation-
1. Sick baby: Sepsis/UTI/Congenital infections/metabolic disorders.
2. Dysmorphism : Downs syndrome/ alagilles syndrome.
3. Examination of eye and fundus must be done-
- Cataract in Galactosemia
- Chorioretinitis in TORCH
- Posterior embryotoxins in alagilles syndrome
- Cherry red spot in Lipid storage disorders
4. Chronic Cholestatis – Pruritis/ irritability/xanthomas
5. Failure to thrive.
*Situation 1: At Secondary Hospital/ Non-Metro situation: Optimal Standards of Treatment in Situations where technology and resources are limited
Clinical Diagnosis:
- Jaundiced child
- Dark urine
- Pale stool
- Hepatomegaly ± splenomegaly
Investigations:
- Hematology: CBC
- Urine routine & microscopy
- Biochemistry:
1. LFT
2. RFT
3. PT/INR
4. RBS - Imaging: USG Abdomen
Treatment:
Standard Operating procedure
- Resuscitation if required,
- Correction of Hypoglycemia
- Administration of Vitamin K (0.3mg/kg parenteral)
- Initiation of antibiotics: if there is clinical or laboratory evidence of infection or sepsis
In Patient : Child needs admission if
- there is clinical evidence of dehydration
- clinical ± laboratory evidence of hypoglycemia
- sepsis or coagulopathy.
- failure to thrive
Out Patient : Baby who is clinically well, feeding well and has no evidence of hypoglycemia or coagulopathy can be investigated as an outpatient
Day Care : No role of day care admission.
Referral criteria : ( All workup at this level must be completed by 48 hours)
1. Any child with neonatal cholestasis syndrome who is > 2 weeks
of age
2. Clinically unwell, poor feeding, poor weight gain
3. Evidence of coagulopathy, hypoglycemia or sepsis
*Situation 2: At Super Specialty Facility in Metro location where higher-end technology is available
Clinical Diagnosis:
- Jaundiced child
- Dark urine
- Pale stool
- Hepatomegaly ± splenomegaly
Investigations: Urgent Investigations
- Blood counts
- LFT
- PT
- Electrolytes
- Blood culture
- Urine culture, Urine microscopy
- Urine reducing substances
- GRBS
- Ascitic tap (if ascites)
Standard LFT are usually abnormal with modestly raised levels of AST, ALT, and alkaline phosphatase. GGT is raised in all cases of cholestasis except in one of the bile acid synthetic defects. Serum albumin does not fall till late. None of the biochemical tests are of deciding value and at best reflect the degree of damage to liver.
Tests directed towards infective and metabolic causes :
Blood Tests
• TORCH, VDRL, Hepatitis B/C, HIV
• T4, TSH
• Serum cortisol
• α l AT levels and phenotype
• Galactose 1 Phosphate Uridyl transferase (to r/o galactosemia)
• Urinary succinyl acetone (to r/o tyrosinemia)
• Cholesterol, triglycerides
• S. iron and ferritin levels (to r/o neonatal hemachromatosis)
Radiology
Role of USG
USG can exclude choledochal cyst, any focal lesions, dilated CBD, anomalies of viscera or
portal hypertension.
Role of Hida Scan
Hepatobiliary scintigraphy, after a 5 day priming with phenobarbitone,is useful. Excretion of the radio-tracer into the gut rules out biliary atresia. However, the converse is not true and absence of gut excretion of radiotracer requires further evaluation.
Role of Liver Biopsy
Liver biopsy is useful in the charecterisation of NCS in some cases. Coagulopathy and ascitis are contraindications for percutaneous liver biopsy.
Treatment
On suspicion of cholestatic liver disease, vitamin-K is started along with supplementation of other fat soluble vitamins (A,D,E,)
Treatable Causes
Medical
- Sepsis
- UTI
- Congenital infections
- Hepatitis
- Galactosemia
- Heriditary fructose intolerance
- Hypothyroidism
- Hypopitaris m
- Tyrosinaemia
Surgical
1. Biliary Atresia
2. Choledochal cyst
3. Spontaneous perforation of bile ducts
4. Inspissation of bile ducts
Supportive Care
Nutrition
- Energy - 125% RDA
- Protein intake -2-3 gm/kg/day in infants (0.5gm/kg/day in hepatic encephalopathy)
- Vitamin A should be supplemented in a dose of 50,000 IU intramuscularly at diagnosis and then 10,000 IU monthly till cholestasis resolves. Avoid hyper- vitaminosis as it can enhance fibrosis.
- Vitamin D should be supplemented in a dose of 30,000 IU intramuscular at diag-nosis and then monthly till cholestasis resolves. If the child has rickets give a dose of 60,000 IU.
- Oral Vitamin E, supplementation (50-200 mg/day) is required to avoid neuro-muscular degeneration, retinal pigmentation and hemolytic anemia.
- Provide Vitamin K 5 mg/day intramuscular/intravenous for first 3 days and then 5 mg weekly. Perform prothombin time (PT) monthly. Administer injectable vitamin K if PT is prolonged.
- Water soluble vitamins and trace elements (2-5) times RDA)
Pruritis
For control of pruritis following agents have been tried :-
1. Phenobarbitone-5mg/kg/day
2. Rifampicin- 1Omg/kg/day
3. Ursodeoxycholic acid-10-20 mg/kg/day
4. Cholestyramine-4- 8gm/kg/day
5. Terfenadine l-3mg/kg/day
6. Carbamazepine
Liver Transplantation
This may remain the only option for infants with decompensated liver disease (ascites and /or encephalopathy) or failed portoenterostomy.
Treatment Plan
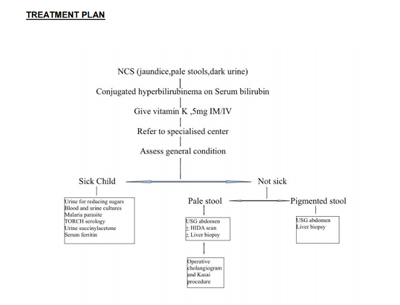
Standard Operating procedure
- In Patient only: May require ICU monitoring
- Referral criteria:
Referral criteria for a specialist centre:
Any case of neonatal cholestasis as defined by above parameters with deranged liver function tests to be referred to tertiary centre for further management.
Child needs to be referred to a specialist pediatric liver unit if,
1. Evidence of progressive liver failure
2. Evidence of complications such as portal
hypertension, SBP, Respiratory distress, pathological
fractures.
3. Failure of Kasai operation
Evidence of liver cirrhosis in biopsy
Jaundice not cleared by 2 months after surgery
4. Considerations for liver transplant
Situation 2:
Referred cases from secondary centres or any newborn with evidence of cholestatis with deranged liver function tests.
Investigation:
| Medical causes | Surgical causes |
| As in situation 1 & Additional investigations Blood culture Urine culture Urine reducing substances Prothrombin time T3/T4/TSH TORCH/VDRL Hep B/C, HIV Metabolic work up | USG Abdomen: Day 5 : Prepare patient for HIDA (Priming with gardenal 3-5 mg/kg) |
Subsequent investigation (if initial workup negative)
- α 1 antitrypsin levels & phenotype
- Galactose 1- P uridyl transferase
- Urinary succinyl acetone
- Cholesterol, Triglycerides
- Serum ferritin, iron
- Radiology
- Rare investigation
Bone marrow aspiration/skin biopsy/muscle biopsy/ serum lactate / pyruvate / ammonia, very long chain fatty acid, urinary organic acids, urinary bile acids, auto immune screen / sweat chloride test.
Treatment:
- Aims of treatment
- Within 7 days of hospitalisation treatment is mandatory.
- Kasai's Portoenterostomy preferably within 60 days of life.
Medical:
Neonatal hepatitis:
2. Bacterial Sepsis/ UTI : antibiotics
3. Malaria : antimalarial
4. Toxoplasmosis & syphilis : specific antibiotics
5. CMV : ganciclovir
6. Herpes : acyclovir
7. Metabolic:
8. Galactosemia : stop lactose milk
9. Fructosemia : withdrawal of fructose containing item
10. Hypothyroidism : Thyroxine
Chronic cholestatis:
- Basically improve nutritional status
- Provide energy (125%RDA)
- Proteins intake 2-3 gm/kg
- MCT diet
- Fat Soluble vitamins (A/D/E/K)
- Pruritis control:
Phenobarbitone / Rifampicin/ UDCA/ Cholestyramine/ Terfenadine/ Carbamazepine
Surgical
| Biliary atresia | Kasai Portoenterostomy |
| Choledochal cyst | Laparatomy |
| Failed Kasai End stage liver disease PFIC | Liver Transplantation |
WHO DOES WHAT? and TIMELINES
Doctor :
- Close and continuous and crucial monitoring of the child
- Planning of tests and treatment
Nurse
- Counseling and support to child and family
- Assisting to treatment
Technician
Resources Required For One Patient / Procedure (Patient Weight 60 KGS)
(Units to be specified for human resources, investigations, drugs and consumables and equipment. Quantity to also be specified)
| Situation | Human resources | Investigations | Drugs & consumables | Equipment |
| 1 | Pediatrician Pediatric Nurse Radiologist Lab. Technician | Vitamin K Antibiotics I.V. Glucose/ Fluids I.V. canula I.v. set | Radiant Warmer Saturation monitor Basic Lab. USG | |
| 2 | Pediatrician with liver intensivists Pediatric surgeon Pediatric anaesthesists Radiologists Gastroenterologis ts Nuclear medicine | Higher antibiotics | NICU Pediatric O.T. Pathologist for biopsy Radiologists Nuclear medicine |
Guidelines by The Ministry of Health and Family Welfare :
- Dr.Sanjay Rao MS,MCh (Paediatric Surgery) Consultant Pediatric Surgeon, Narayana Hrudayalaya, Bangalore
- Dr.Rajiv Aggarwal, MD Consultant Pediatrician and Neonatologist Narayana Hrudayalaya, Bangalore
Assisted by:
1. Dr.Vinay C
2. Dr. Zameer K Both from Department of Pediatric Surgery Narayana Hrudayalaya,Bangalore

Disclaimer: This site is primarily intended for healthcare professionals. Any content/information on this website does not replace the advice of medical and/or health professionals and should not be construed as medical/diagnostic advice/endorsement or prescription. Use of this site is subject to our terms of use, privacy policy, advertisement policy. © 2020 Minerva Medical Treatment Pvt Ltd