- Home
- Editorial
- News
- Practice Guidelines
- Anesthesiology Guidelines
- Cancer Guidelines
- Cardiac Sciences Guidelines
- Critical Care Guidelines
- Dentistry Guidelines
- Dermatology Guidelines
- Diabetes and Endo Guidelines
- Diagnostics Guidelines
- ENT Guidelines
- Featured Practice Guidelines
- Gastroenterology Guidelines
- Geriatrics Guidelines
- Medicine Guidelines
- Nephrology Guidelines
- Neurosciences Guidelines
- Obs and Gynae Guidelines
- Ophthalmology Guidelines
- Orthopaedics Guidelines
- Paediatrics Guidelines
- Psychiatry Guidelines
- Pulmonology Guidelines
- Radiology Guidelines
- Surgery Guidelines
- Urology Guidelines
Need for caution-Dexmedetomidine clearance low during Cardiopulmonary Bypass in Kids
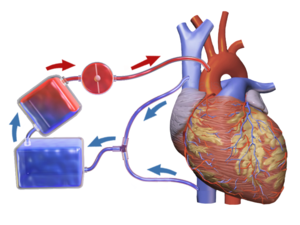
Cardiopulmonary bypass (CPB) is associated with lower clearance of Dexmedetomidine during CPB procedure in infants and young children compared to those not undergoing CPB, according to a study published in the Anesthesia & Analgesia. This, therefore, calls for a caution and alteration of dosing of Dexmedetomidine after Cardiopulmonary bypass.
Dexmedetomidine is increasingly used off-label in infants and children with the cardiac disease during cardiopulmonary bypass (CPB) and in the postoperative period. Cardiopulmonary bypass (CPB) is a technique that temporarily takes over the function of the heart and lungs during surgery maintaining the circulation of blood and the oxygen content of the patient's body. The CPB pump itself is often referred to as a heart-lung machine
Kanecia O. Zimmerman conducted the study to characterize the effect of CPB on dexmedetomidine clearance (CL) and volume of distribution (V) in infants and young children, characterize tolerance and sedation in patients receiving dexmedetomidine; and identify preliminary dosing recommendations for infants and children undergoing CPB.
The researchers hypothesized that clearance would decrease, and volume of distribution would increase during CPB compared to pre- or post-CPB states.
The researchers conducted an open-label, single-centred, opportunistic pharmacokinetics (PK) and safety study of dexmedetomidine in patients ≤36 months of age administered dexmedetomidine per standard of care via continuous infusion.
The authors analyzed dexmedetomidine pharmacokinetics, PK data using standard nonlinear mixed effects modelling with NONMEM software and compared model-estimated PK parameters to those from historical patients receiving dexmedetomidine before anesthesia for urologic, lower abdominal, or plastic surgery; after low-risk cardiac or craniofacial surgery or during bronchoscopy or nuclear magnetic resonance imaging.
The authors investigated the influence of CPB-related factors on pharmacokinetics,PK estimates and used the final model to simulate dosing recommendations, targeting a plasma concentration previously associated with safety and efficacy (0.6 ng/mL). They collected 213 dexmedetomidine plasma samples from 18 patients. Patients had a median age of 3.3 months and underwent CPB for 161 minutes (63–394 minutes).
Read Also: Guideline on Temperature Management during cardiopulmonary bypass
Key study findings:
- A clearance of 13.4 L/h/70 kg was estimated CPB, compared to 42.1 L/h/70 kg in the historical patients.
- No specific CPB-related factor had a statistically significant effect on PK.
- A loading dose of 0.7 µg/kg over 10 minutes before CPB, followed by maintenance infusions through CPB of 0.2 or 0.25 µg/kg/h in infants with postmenstrual ages of 42 or 92 weeks, respectively, maintained targeted concentrations.
- No association was identified between dexmedetomidine exposure and selected adverse events.
The researchers concluded that CPB is associated with lower clearance during CPB in infants and young children compared to those not undergoing CPB. But further study should more closely investigate CPB-related factors that may influence CL.
For reference log on to 10.1213/ANE.0000000000003700

Disclaimer: This site is primarily intended for healthcare professionals. Any content/information on this website does not replace the advice of medical and/or health professionals and should not be construed as medical/diagnostic advice/endorsement or prescription. Use of this site is subject to our terms of use, privacy policy, advertisement policy. © 2020 Minerva Medical Treatment Pvt Ltd