- Home
- Editorial
- News
- Practice Guidelines
- Anesthesiology Guidelines
- Cancer Guidelines
- Cardiac Sciences Guidelines
- Critical Care Guidelines
- Dentistry Guidelines
- Dermatology Guidelines
- Diabetes and Endo Guidelines
- Diagnostics Guidelines
- ENT Guidelines
- Featured Practice Guidelines
- Gastroenterology Guidelines
- Geriatrics Guidelines
- Medicine Guidelines
- Nephrology Guidelines
- Neurosciences Guidelines
- Obs and Gynae Guidelines
- Ophthalmology Guidelines
- Orthopaedics Guidelines
- Paediatrics Guidelines
- Psychiatry Guidelines
- Pulmonology Guidelines
- Radiology Guidelines
- Surgery Guidelines
- Urology Guidelines
More Corneal aberrations observed in Down Syndrome
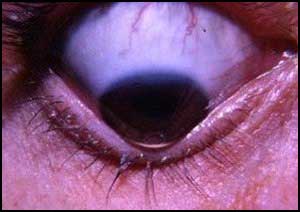
Patients with Down Syndrome have steeper and thinner corneas and more corneal aberrations than those without genetic alterations and normal corneas suggest a study recently published in the journal JAMA Opthalmology.
According to the literature review, corneal morphologic characteristics compatible with keratoconus are present in a high percentage of patients with Down syndrome (DS), which can cause serious visual impairment in this group of patients, suggesting the need to perform a detailed examination of the anterior segment.
Keratoconus (KC) is a disorder of the eye which results in progressive thinning of the cornea. This may result in blurry vision, double vision, nearsightedness, astigmatism, and light sensitivity.
Jorge L. Alio and his associates conducted a multicenter case-control study to characterize the abnormal features of the cornea in patients with Down Syndrome and compare these with a control group.
The study included 321 eyes of 217 participants from 2 groups: 112 participants in the DS group and 105 healthy participants in the control group.
Patients were evaluated using a Placido disc/Scheimpflug camera topographer (Sirius, CSO). Visual, refractive and anterior, and posterior corneal characteristics were assessed and compared in both groups.
The main outcome measured was the incidence of corneal morphologic irregularities similar to keratoconus in patients with DS.
Read Also: “Down Syndrome Screening in India: Are we there yet?”
The study found that clinical assessment of corneal topography showed 71.3% of patients in the Down Syndrome group showed characteristics compatible with keratoconus. Differences were found in steepest keratometry of 47.35 diopters (D) in patients with Down Syndrome vs 43.70 D in control individuals and in corneal pachymetry of 503 μm in patients with DS vs 545 μm in control individuals.
The authors concluded that the findings suggest a detailed corneal study to be considered in patients with Down Syndrome to detect keratoconus so as to have an appropriate treatment to avoid serious visual impairment.
For more reference log on to
https://jamanetwork.com/journals/jamaophthalmology/article-abstract/2685542

Disclaimer: This site is primarily intended for healthcare professionals. Any content/information on this website does not replace the advice of medical and/or health professionals and should not be construed as medical/diagnostic advice/endorsement or prescription. Use of this site is subject to our terms of use, privacy policy, advertisement policy. © 2020 Minerva Medical Treatment Pvt Ltd