- Home
- Editorial
- News
- Practice Guidelines
- Anesthesiology Guidelines
- Cancer Guidelines
- Cardiac Sciences Guidelines
- Critical Care Guidelines
- Dentistry Guidelines
- Dermatology Guidelines
- Diabetes and Endo Guidelines
- Diagnostics Guidelines
- ENT Guidelines
- Featured Practice Guidelines
- Gastroenterology Guidelines
- Geriatrics Guidelines
- Medicine Guidelines
- Nephrology Guidelines
- Neurosciences Guidelines
- Obs and Gynae Guidelines
- Ophthalmology Guidelines
- Orthopaedics Guidelines
- Paediatrics Guidelines
- Psychiatry Guidelines
- Pulmonology Guidelines
- Radiology Guidelines
- Surgery Guidelines
- Urology Guidelines
Management of Community - onset Acute Undifferentiated Fever in Adults- ICMR Guidelines

Acute undifferentiated fever (AUF) is a temporary febrile illness accompanied by non-specific symptoms. Previous studies in Asia have primarily focused on delineating specific agent(s) causing AUF. None were designed to assess the frequency of undiagnosed cases.
Indian Council of Medical Research, Department of Health Research has issued the ICMR treatment Guidelines for Management of Community - onset Acute Undifferentiated Fever in Adults. Following are the major recommendations :
Outline:
The purpose of the guidelines is to ensure appropriate antimicrobial treatment while at the same time limiting the inappropriate use of antibiotics in the management of infections by addressing issues like antibiotic selection, dosing, route, duration, adverse drug events, and cost and prevention of unintended collateral damage.
Some general principles:
- Antibiotic use will need to be classified with respect to type (high- and low-risk) and the patient’s place in the treatment pathway (untreated, treated, and posttreatment).
- The choice of medication may vary depending on differences in the case mix of patients, various drugs (of same or different class) listed in formulary or clinical practice guidelines already in place at different institutions in similar patient care locations.
- Timely use of diagnostic tests or documentation of symptoms supporting the presence of infection would be best. Cultures (two sets of blood cultures and other appropriate samples as clinically indicated e.g. normally sterile body fluids, deep pus etc.) should be taken before starting empiric antibiotic treatment.
- Empiric antibiotic treatment for common infections should be limited to conditions where early initiation of antibiotics has been shown to be beneficial, eg severe sepsis and septic shock, acute bacterial meningitis, community acquired pneumonia, necrotizing fasciitis, etc.
- Reassessment of the situation within 48 hours based on the test results and examination of the patient is required. If needed, the drug's dosage and duration can be adjusted or the antibiotic regimen should be de-escalated (to the narrowest spectrum, least toxic and least expensive antibiotic) based upon patient response and culture and susceptibility reports.
Case Definition
- Previously healthy (non-immunosuppressed) community (urban or rural) dwelling adult (ages 19-64 yrs) reporting no previous medical illness or recent hospitalization (in the preceding 30 days) presenting with acute onset of fever > 38.3° C (101.0° F) for > 2 days and lasting up to 14 days and having received no specific treatment for this current illness with antimalarials or antibiotics.
- Seen in ambulatory care settings at the primary level (PHC), doctor’s office/clinic, emergency room in a community Hospital, including referrals from primary health care or community physicians.
- With history of no localizing symptoms (except accompaniments of fever such as – chills, headaches, retro-orbital pain, myalgia, malaise, nausea or vomiting). On examination found to have normal vital signs (excepting fever) and lacking organ or system specific physical signs.*
* A complete and thorough physical examination is mandatory. Record of vital signs is essential. A search is required for hidden foci such as throat examination, sinus tenderness, renal or hepatic tenderness, heart murmurs, chest examination, lymph nodes and splenomegaly. Fundus examination (if headache or bleeding tendency) and examination of the skin for eschar and petechiae or purpura must be made.
Common pathogens causing “tropical fevers”, “seasonal fevers” “endemic /epidemic /outbreak fever”, “monsoon fever”:
- Suspect malaria in all cases of acute undifferentiated fever (there are no key differentiating features between this and other causes (see below). Despite historical claims, fever patterns are not especially helpful in establishing a specific diagnosis. Malaria is especially to be suspected after a visit to high malaria endemic zone.
- Viruses cause febrile illness or specific viral “influenza like- illness” (with mild sore throat and cough).
- If rash or exanthema is present without drug exposure (rule out drug allergy), consider mononucleosis syndrome (EBV, CMV, HIV) or an exanthematous viral illness (measles, rubella, etc).
- Primary or secondary dengue (sometimes accompanied by maculo-papular rash or polyarthralgia). Tourniquet test may be inappropriate as a general discriminating test without hemorrhagic manifestations or the shock syndrome. Consider hemorrhagic fever with two or more hemorrhagic symptoms - hemorrhagic or purpuric rash, epistaxis, conjunctival hemorrhage, bleeding gums, bleeding at puncture sites, hematuria, hematemesis, hemoptysis, blood in stools .
- Scrub typhus or murine typhus may present with skin eschar, regional lymphadenopathy, and maculopapular rash.
- Leptospirosis can present with conjunctival suffusion, muscle tenderness and jaundice (ask for flood water or sewage exposure)
- Typhoid (continuous fever and splenomegaly are clues)
- Community acquired secondary bacteremia: Primary source may be occult. In most instances it is either from an underlying pneumonia, intra-abdominal infection or urosepsis. Symptoms related to these systems may not be manifest, especially in the elderly.
- Hepatitis A or E (fever usually subsides with onset of jaundice)
- Chikungunya presents with arthralgia /polyarthritis.
- Consider rheumatic fever caused by Group A beta hemolytic streptococci if there is migratory arthritis with preceding significant “sore throat”.
- Tuberculosis should be considered in any patient with prolonged undifferentiated fever, especially if there is weight loss or night sweats.
Diagnostic Investigations (where facilities are available)
- One blood smear and/or RDT at least is required for malarial parasite detection (repeat blood smear once more if initial smear is negative in an endemic region).
- Complete blood count : Anemia, leucopenia /leukocytosis, elevated hematocrit or thrombocytopenia (dengue, leptospirosis)
- Diagnostic blood cultures for aerobic bacteria (at least two) to be drawn prior to start of empiric antibiotics
- Liver enzymes and bilirubin
- Urinalysis – white blood cells, proteinuria and hematuria.
- Chest roentgenogram (if chest findings are present, to rule out early pneumonia or TB)
- Ultrasonography of abdomen if fever persists to rule out hepatic, renal or intraabdominal sources of infection.
- Within 96 hours of onset of fever, antigen based serological tests are likely to be positive whereas antibody tests are generally positive after at least 5-7 days of illness.
- Dengue rapid NS1 antigen and IgM Combo test (SD bioline, USA)
- Scrub typhus IgM ELISA (In Bios, USA),
- Leptospira IgM ELISA (Alere, Australia),
Prevalent AMR status in common pathogens
Malaria: P. vivax is susceptible to chloroquine which remains the drug of choice. P. falciparum resistance to chloroquine is seen in at least 25% of cases nationwide, and therefore artemisinin-based combination therapies (ACT) should be the first line treatment for P. falciparum malaria and where species is unclear. Artemesinin (especially oral) monotherapy should be strongly discouraged as it leads to resistance this class.
Typhoid fever: Quinolone resistance is increasing and is as high as 69% for Salmonella Typhi and 23% for S. Paratyphi A. Resistance rates are low for co-trimoxazole, chloramphenicol and third generation cephalosporins. Though sensitivity testing is not validated for azithromycin, response is good in most clinical studies. However defervescence times are significantly longer with third generation cephalosporins compared with other classes and bone marrow depression is a concern with chloramphenicol.
Table 1. Salmonella Typhi ICMR AMR Data 2014
| AMA | PGIMER, Chandigarh ‘n’ 109 No. R (%) | AIIMS, New Delhi ‘n’ 22 No. R | CMC, Vellore ‘n’ 71 No. R (%) | JIPMER, Puducherry ‘n’ 7 No. R | National ‘n’ 209 No.R %) |
| Ampicillin | 9 (8.3) | 0 | 2 (2.8) | 0 | 11 (5.3) |
| Cefixime | 0 (0) | 0 | 0 (0) | 0 | 0 (0) |
| Ceftriaxone | 0 (0) | 0 | 0 (0) | 0 | 0 (0) |
| Chloramphenicol | 3 (2.8) | 0 | 1 (1.4) | 0 | 4 (1.9) |
| Ciprofloxacin | 56 (51.4) | 15 | 67 (94.4) | 7 | 145 (69.4) |
| Trimethoprim-sulphamethoxazole | 0 (0) | 0 | 3 (4.2) | 0 | 3 (1.4) |
Note : If No. Tested is ≥30, No. R (%) given. If No. tested <30, only No. R given.
Table 2. Salmonella Paratyphi A ICMR AMR Data 2014
| AMA | PGIMER, Chandigarh ‘n’ 2 No. R | AIIMS, New Delhi ‘n’ 6 No. R | CMC, Vellore ‘n’ 14 No. R | JIPMER, Puducherry ‘n’ 7 No. R | National ‘n’ 29 No. R |
| Ampicillin | 1 | 0 | 0 | 1 | 2 |
| Cefixime | 0 | 0 | 0 | 0 | 0 |
| Ceftriaxone | 0 | 0 | 0 | 0 | 0 |
| Chloramphenicol | 0 | 0 | 1 | 0 | 1 |
| Ciprofloxacin | 1 | 2 | 14 | 6 | 23 |
| Trimethoprim-sulphamethoxazole | 0 | 0 | 0 | 0 | 0 |
Note : If No. Tested is ≥30, No. R (%) given. If No. tested <30, only No. R given.
Gram positive organisms: Community acquired organisms such as S. aureus are usually susceptible to methicillin i.e. standard anti staphylococcal drugs such as cloxacillin and first cephalosporins may be used. Penicillin still remains the drug of choice for pneumococcal infection.
Table 3. Staphylococcus aureus ICMR AMR Data 2014
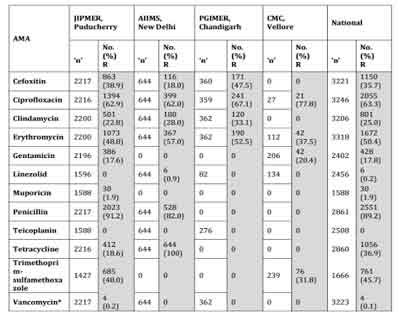
*The 4 numbers listed as Vancomycin Resistant (R) are VISA isolates.
No VRSA was isolated during the year 2014 at JIPMER.
Cefoxitin : Surrogate marker for Methicillin.
Gram negative organisms: On the other hand, there is increasing resistance among Enterobacteriaceae (E. coli and Klebsiella) to both quinolones (up to 80%) and third generation cephalosporins (up to 75% on account of ESBL production). Initial empiric therapy for infections caused by these organisms {pyelonephritis, severe intraabdominal infections (IAI), etc} should be with an agent active against ESBL producers eg beta-lactam/beta-lactamase inhibitors or with a carbapenem for more severely ill patients.
Table 4 . Enterobacteriaceae isolates from blood. ICMR AMR data 2014.
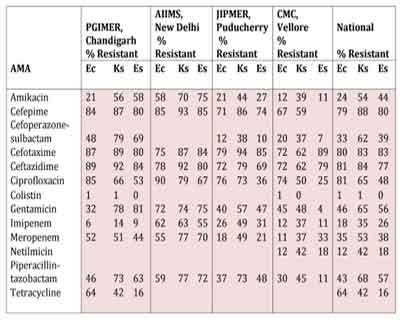
Note : Ec : Escherichia coli; Ks : Klebsiella spp.; Es : Enterobacter spp
7. Principles of empiric therapy:
- Supportive: Acetaminophen 500 or 650 mg every 6 hours round the clock is advisable, accompanied by tepid sponging for fever or with chills >103 F. Replace fluid and electrolytes as required.
- No antibiotics are required for the majority of patients with acute febrile illness without an obvious clinical diagnosis.
- Always draw two sets of blood cultures before start of empiric antibacterial therapy.
- Start antibiotics for a presumed bacterial infection promptly, but adjust the drug's dosage and duration, switch to a new drug, or end antibiotic therapy when results do not support or justify the need to continue.
- Reassess the situation within 48 hours based on test results and patient status.
- Corticosteroids are not recommended in the treatment of acute undifferentiated fever.
- In patients with fever and thrombocytopenia, platelet transfusions are not recommended in general.
- Consider platelet transfusion when platelet counts are <10,000 cumm or in the presence of clinical bleeding in cases of dengue hemorrhagic fever.
- Empirical treatment with doxycycline for patients with undifferentiated fever and negative rapid diagnostic tests for malaria and dengue is an option for the clinician. (Lancet Glob Health. 2013 Jul;1(3):e46-e54.)
Figure 1 Protocol for the management of adult patients with acute undifferentiated fever.
(Int J Emerg Med. 2011; 4: 57)
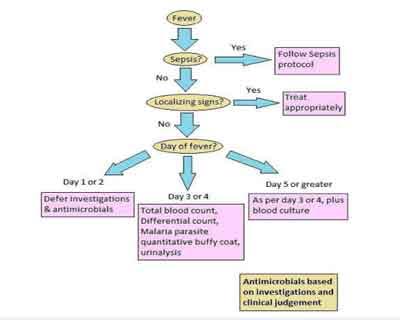
Table 5 Antimicrobial Choice for Disease conditions
| Type of disease | Organisms | Initial Treatment/ Preferred | Alternatives | Comments | |
| 1. | Typhoid fever | Salmonella Typhi, Salmonella Paratyphi A | Oral: cotrimoxazole (1ds tab bd) or azithromycin (10 mg/kg/day) Parenteral: ceftriaxone 2 g IV od | Cefixime (20 mg/kg/day) or chloramphenico l 500 mg qid or ciprofloxacin 750 mg bd | Change empiric regimen based on susceptibility testing. Duration of treatment: 10-14 days. |
| 2. | Empiric therapy of suspected Gram positive infections | S.pneumoniae, Streptococcus pyogenes, Staphylococcu s aureus | Cefazolin 2 g IV q8h or Cloxacillin 2 g IV q6h | Amoxicilinclavulanate 1.2 g IV q8h or Penicillin G 20 laks IV q4h (if S.aureus excluded) or Vancomycin (if anaphylactic penicillin allergy or MRSA clinically possible) | Adjust regimen after receipt of culture and susceptibility data. Duration of treatment will depend on final diagnosis. |
| 3. | Empiric therapy for suspected Gram negative infections (eg pyelonephrit is or intraabdominal infections) | E. coli, Klebsiella pneumoniae, anaerobes especially Bacteroides sp in IAI | Piperacillinta zobactam 4.5 g IV q6h or Cefoperazone -sulbactam 3 g IV q12h | Imipenem 1 g IV q8h or Meropenem 1 g IV q8h or Ertapenem 1 g IV od (carbapenems preferred for more seriously ill patients) | Separate anaerobic coverage unnecessary for IAI, when using BL-BLIs or carbapenems. De-escalate to ciprofloxacin, cotrimoxazole or third generation cephalosporin if isolate is sensitive. Duration of treatment: 10-14 days for pyelonephritis, 4-7 days for IAI. |
| 4. | Rickettsial infections | Orientia tsutsugamushi , Rickettsia conori | Doxycycline 100 mg po or IV bd | Azithromycin 500 mg po or IV od, chloramphenico l 500mg qid | Duration of treatment: 7 days |
| 5. | Leptospirosi s | Leptospira sp | Penicillin G 20 laks IV q4h or doxycycline 100 mg po or IV bd | Ceftriaxone 2 g IV od | Duration of treatment: 7 days |
| 6. | Vivax malaria | P.vivax | Chloroquine 25 mg/kg body weight divided over three days i.e. 10 mg/kg on day 1, 10 mg/kg on day 2 and 5 mg/kg on day 3. | Artemetherlumefantrine (1 tab bd for 3 days) | Followed by primaquine (0.25 mg/kg daily for 14 days) |
| 7. | Falciparum malaria | P. falciparum | Artesunate 4 mg/kg body weight daily for 3 days Plus Sulfadoxine (25 mg/kg body weight) and Pyrimethami ne (1.25 mg/kg body weight) on first day. | Artemetherlumefantrine (1 tab bd for 3 days) | Followed by primaquine single dose (0.75 mg/kg). All mixed infections should be treated with full course of ACT and primaquine 0.25 mg per kg daily for 14 days. |
You can read the full Guideline by clicking on the link :
Dr Soumya Swaminathan, Director General, Indian Council of Medical Research Secretary, Department of Health Research

Disclaimer: This site is primarily intended for healthcare professionals. Any content/information on this website does not replace the advice of medical and/or health professionals and should not be construed as medical/diagnostic advice/endorsement or prescription. Use of this site is subject to our terms of use, privacy policy, advertisement policy. © 2020 Minerva Medical Treatment Pvt Ltd