- Home
- Editorial
- News
- Practice Guidelines
- Anesthesiology Guidelines
- Cancer Guidelines
- Cardiac Sciences Guidelines
- Critical Care Guidelines
- Dentistry Guidelines
- Dermatology Guidelines
- Diabetes and Endo Guidelines
- Diagnostics Guidelines
- ENT Guidelines
- Featured Practice Guidelines
- Gastroenterology Guidelines
- Geriatrics Guidelines
- Medicine Guidelines
- Nephrology Guidelines
- Neurosciences Guidelines
- Obs and Gynae Guidelines
- Ophthalmology Guidelines
- Orthopaedics Guidelines
- Paediatrics Guidelines
- Psychiatry Guidelines
- Pulmonology Guidelines
- Radiology Guidelines
- Surgery Guidelines
- Urology Guidelines
Management of carpal tunnel syndrome : American Academy of Orthopaedic Surgeons guidelines
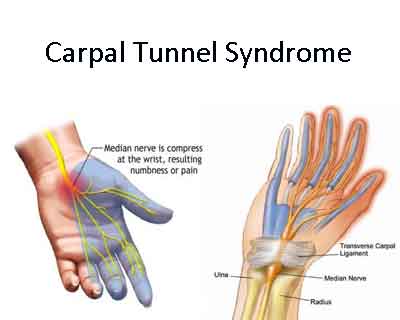
Carpal Tunnel Syndrome is a symptomatic compression neuropathy of the median nerve at the level of the wrist, characterized physiologically by evidence of increased pressure within the carpal tunnel and decreased function of the nerve at that level. Carpal Tunnel Syndrome can be caused by many different diseases, conditions and events. It is characterized by patients as producing numbness, tingling, hand and arm pain and muscle dysfunction. The disorder is not restricted by age, gender, ethnicity, or occupation and is associated with or caused by systemic disease and local mechanical and disease factors.
In 2016, American Academy of Orthopedic Surgeons came out with the guidelines on Management of Carpel Tunnel Syndrome- Evidence based clinical practice guidelines
Following are its major recommendations:-
Observation
Strong evidence supports thenar atrophy is strongly associated with ruling-in carpal tunnel syndrome (CTS), but poorly associated with ruling-out CTS.
Physical Signs
Strong evidence supports not using the Phalen test, Tinel sign, Flick sign, or Upper limb neurodynamic/nerve tension test (ULNT) criterion A/B as independent physical examination maneuvers to diagnose carpal tunnel syndrome, because alone, each has a poor or weak association with ruling-in or ruling-out CTS.
Maneuvers
Moderate evidence supports not using the following as independent physical examination maneuvers to diagnose CTS, because alone, each has a poor or weak association with ruling-in or ruling-out CTS:
- Carpal compression test
- Reverse Phalen test
- Thenar weakness or thumb abduction weakness or abductor pollicis brevis manual muscle testing
- 2-point discrimination
- Semmes-Weinstein monofilament test
- CTS-relief maneuver (CTS-RM)
- Pin prick sensory deficit; thumb or index or middle finger
- ULNT Criterion C
- Tethered median nerve stress test
- Vibration perception- tuning fork
- Scratch collapse test
- Luthy sign
- Pinwheel
History Interview Topics
Moderate evidence supports not using the following as independent history interview topics to diagnose CTS, because alone, each has a poor or weak association with ruling-in or ruling-out CTS:
- Sex/gender
- Ethnicity
- Bilateral symptoms
- Diabetes mellitus
- Worsening symptoms at night
- Duration of symptoms
- Patient localization of symptoms
- Hand dominance
- Symptomatic limb
- Age
- Body mass index ()
Patient Reported Numbness or Pain
Limited evidence supports that patients who do not report frequent numbness or pain might not have CTS.
Hand-held Nerve Conduction Study (NCS)
Limited evidence supports that a hand-held NCS device might be used for the diagnosis of CTS.
Magnetic Resonance Imaging (MRI)
Moderate evidence supports not routinely using MRI for the diagnosis of CTS.
Diagnostic Ultrasound
Limited evidence supports not routinely using ultrasound for the diagnosis of CTS.
Diagnostic Scales
Moderate evidence supports that diagnostic questionnaires and/or electrodiagnostic studies could be used to aid the diagnosis of CTS.
Increased Risk of CTS
- Strong evidence supports that BMI and high hand/wrist repetition rate are associated with the increased risk of developing CTS.
- Peri-menopausal
- Wrist ratio/index
- Rheumatoid arthritis
- Psychosocial factors
- Distal upper extremity tendinopathies
- Gardening
- American Conference of Governmental Industrial Hygienists Hand Activity Level at or above threshold
- Assembly line work
- Computer work
- Vibration
- Tendinitis
- Workplace forceful grip/exertionModerate evidence supports that the following factors are associated with the increased risk of developing CTS:
- Dialysis
- Fibromyalgia
- Varicosis
- Distal radius fractureLimited evidence supports that the following factors are associated with the increased risk of developing CTS:
Decreased Risk of CTS
Moderate evidence supports that physical activity/exercise is associated with the decreased risk of developing CTS.
Factors Showing No Associated Risk of CTS
- Moderate evidence supports that the use of oral contraception and female hormone replacement therapy (HRT) are not associated with increased or decreased risk of developing CTS.
- Limited evidence supports that race/ethnicity and female education level are not associated with increased or decreased risk of developing CTS.
Factors Showing Conflicting Risk of CTS
Limited evidence supports that the following factors have conflicting results regarding the development of CTS:
- Diabetes
- Age
- Gender/sex
- Genetics
- Comorbid drug use
- Smoking
- Wrist bending
- Workplace
Immobilization
Strong evidence supports that the use of immobilization (brace/splint/orthosis) should improve patient reported outcomes.
Steroid Injections
Strong evidence supports that the use of steroid (methylprednisolone) injection should improve patient reported outcomes.
Magnet Therapy
Strong evidence supports not using magnet therapy for the treatment of CTS.
Oral Treatments
Moderate evidence supports no benefit of oral treatments (diuretic, gabapentin, astaxanthin capsules, non-steroidal anti-inflammatory drugs [NSAIDs], or pyridoxine) compared to placebo.
Oral Steroids
Moderate evidence supports that oral steroids could improve patient reported outcomes as compared to placebo.
Ketoprofen Phonophoresis
Moderate evidence supports that ketoprofen phonophoresis could provide reduction in pain compared to placebo.
Therapeutic Ultrasound
Limited evidence supports that therapeutic ultrasound might be effective compared to placebo.
Laser Therapy
Limited evidence supports that laser therapy might be effective compared to placebo.
Surgical Release Location
Strong evidence supports that surgical release of the transverse carpal ligament should relieve symptoms and improve function.
Surgical Release Procedure
Limited evidence supports that if surgery is chosen, a practitioner might consider using endoscopic carpal tunnel release based on possible short term benefits.
Surgical Versus Nonoperative
Strong evidence supports that surgical treatment of carpal tunnel syndrome should have a greater treatment benefit at 6 and 12 months as compared to splinting, NSAIDs/therapy, and a single steroid injection.
Adjunctive Techniques
Moderate evidence supports that there is no benefit to routine inclusion of the following adjunctive techniques: epineurotomy, neurolysis, flexor tenosynovectomy, and lengthening/reconstruction of the flexor retinaculum (transverse carpal ligament).
Bilateral Versus Staged Carpal Tunnel Release
Limited evidence supports that simultaneous bilateral or staged endoscopic carpal tunnel release might be performed based on patient and surgeon preference. No evidence meeting the inclusion criteria was found addressing bilateral simultaneous open carpal tunnel release.
Local Versus Intravenous (IV) Regional Anesthesia
Limited evidence supports the use of local anesthesia rather than IV regional anesthesia (bier block) because it might offer longer pain relief after carpal tunnel release; no evidence meeting the inclusion criteria was found comparing general anesthesia to either regional or local anesthesia for carpal tunnel surgery.
Buffered Versus Plain Lidocaine
Moderate evidence supports the use of buffered lidocaine rather than plain lidocaine for local anesthesia because it could result in less injection pain.
Aspirin Use
Limited evidence supports that the patient might continue the use of aspirin perioperatively; no evidence meeting the inclusion criteria addressed other anticoagulants.
Preoperative Antibiotics
Limited evidence supports that there is no benefit for routine use of prophylactic antibiotics prior to carpal tunnel release because there is no demonstrated reduction in postoperative surgical site infection.
Supervised Versus Home Therapy
Moderate evidence supports no additional benefit to routine supervised therapy over home programs in the immediate postoperative period. No evidence meeting the inclusion criteria was found comparing the potential benefit of exercise versus no exercise after surgery.
Postoperative Immobilization
Strong evidence supports no benefit to routine postoperative immobilization after carpal tunnel release.
For detailed guidelines click on the following link:
http://www.aaos.org/uploadedFiles/PreProduction/Quality/Guidelines_and_Reviews/guidelines/CTS CPG_6 7 2016.pdf

Disclaimer: This site is primarily intended for healthcare professionals. Any content/information on this website does not replace the advice of medical and/or health professionals and should not be construed as medical/diagnostic advice/endorsement or prescription. Use of this site is subject to our terms of use, privacy policy, advertisement policy. © 2020 Minerva Medical Treatment Pvt Ltd