- Home
- Editorial
- News
- Practice Guidelines
- Anesthesiology Guidelines
- Cancer Guidelines
- Cardiac Sciences Guidelines
- Critical Care Guidelines
- Dentistry Guidelines
- Dermatology Guidelines
- Diabetes and Endo Guidelines
- Diagnostics Guidelines
- ENT Guidelines
- Featured Practice Guidelines
- Gastroenterology Guidelines
- Geriatrics Guidelines
- Medicine Guidelines
- Nephrology Guidelines
- Neurosciences Guidelines
- Obs and Gynae Guidelines
- Ophthalmology Guidelines
- Orthopaedics Guidelines
- Paediatrics Guidelines
- Psychiatry Guidelines
- Pulmonology Guidelines
- Radiology Guidelines
- Surgery Guidelines
- Urology Guidelines
Ixekizumab shows promise in resistant ankylosing spondylitis
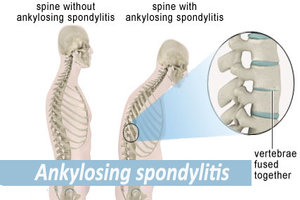
Ixekizumab shows promise in resistant ankylosing spondylitis according to a new study.
The study published in the journal Arthritis & Rheumatology reports that Ixekizumab treatment for 16 weeks in patients with ankylosing spondylitis (AS) who do not achieve adequate disease control or symptom relief from tumor necrosis factor (TNF) inhibitors yielded rapid and significant improvements in the signs and symptoms of the disease. The findings were presented at the American College of Rheumatology's annual meeting in Chicago.
Atul Deodhar and associates conducted a randomized, double-blind, placebo-controlled Phase 3 clinical trial to investigate the efficacy and safety of ixekizumab in patients with active radiographic axial spondyloarthritis (r‐axSpA) and prior inadequate response or intolerance to 1 or 2 TNF inhibitors (TNFi).
Read Also: FDA Approves Golimumab for Psoriatic Arthritis, Ankylosing Spondylitis
Ixekizumab is a high-affinity monoclonal antibody that selectively targets IL-17A, in patients with AS who experienced a previous inadequate response or intolerance to TNF inhibitors. The trial included 316 patients who were randomized 1:1:1 to placebo, ixekizumab every 2 weeks (IXEQ2W), and ixekizumab every 4 weeks (IXEQ4W), with an 80-mg or 160-mg starting dose.
The key study findings included are:
- At Week 16, significantly higher proportions of IXEQ2W patients achieved ASAS40 versus placebo with statistically significant differences reported as early as Week 1 with ixekizumab treatment.
- Statistically significant improvements in disease activity, function, quality of life, and spinal MRI inflammation were observed with 16 weeks of ixekizumab treatment versus placebo.
- Treatment‐emergent adverse events (AE) with ixekizumab treatment were more frequent than with placebo.
- Serious AEs were similar across treatment arms. One death was reported (IXEQ2W).
"Many people with this chronic, debilitating disease are still searching for an effective treatment. These positive results provide support for ixekizumab as a potential treatment option for patients with AS, including those who have had an inadequate response to treatment with TNF inhibitors, a difficult-to-treat population," said Dr. Deodhar.
Read Also: Alarming rise in Ankylosing Spondylitis amongst young Indian workforce
Ankylosing spondylitis (AS) is a chronic inflammatory disease characterized by back pain and sacroiliac-joint damage with tumor necrosis factor (TNF) inhibitors being the currently recommended treatment for AS but unfortunately, 30 to 40 percent of patients with AS do not achieve adequate disease control with the existing recommended treatment.
In addition, some patients may not be eligible to receive the recommended treatment due to contraindications. The cytokine IL-17 is thought to play a role in the development of AS, and IL-17 inhibitors are effective in some patients, but they have not been evaluated exclusively in patients who have not experienced symptom relief with TNF inhibitors.
For reference log on to https://doi.org/10.1002/art.40753

Disclaimer: This site is primarily intended for healthcare professionals. Any content/information on this website does not replace the advice of medical and/or health professionals and should not be construed as medical/diagnostic advice/endorsement or prescription. Use of this site is subject to our terms of use, privacy policy, advertisement policy. © 2020 Minerva Medical Treatment Pvt Ltd