- Home
- Editorial
- News
- Practice Guidelines
- Anesthesiology Guidelines
- Cancer Guidelines
- Cardiac Sciences Guidelines
- Critical Care Guidelines
- Dentistry Guidelines
- Dermatology Guidelines
- Diabetes and Endo Guidelines
- Diagnostics Guidelines
- ENT Guidelines
- Featured Practice Guidelines
- Gastroenterology Guidelines
- Geriatrics Guidelines
- Medicine Guidelines
- Nephrology Guidelines
- Neurosciences Guidelines
- Obs and Gynae Guidelines
- Ophthalmology Guidelines
- Orthopaedics Guidelines
- Paediatrics Guidelines
- Psychiatry Guidelines
- Pulmonology Guidelines
- Radiology Guidelines
- Surgery Guidelines
- Urology Guidelines
Onyx one stent with one month of DAPT non inferior to biofreedom stent in high risk patients
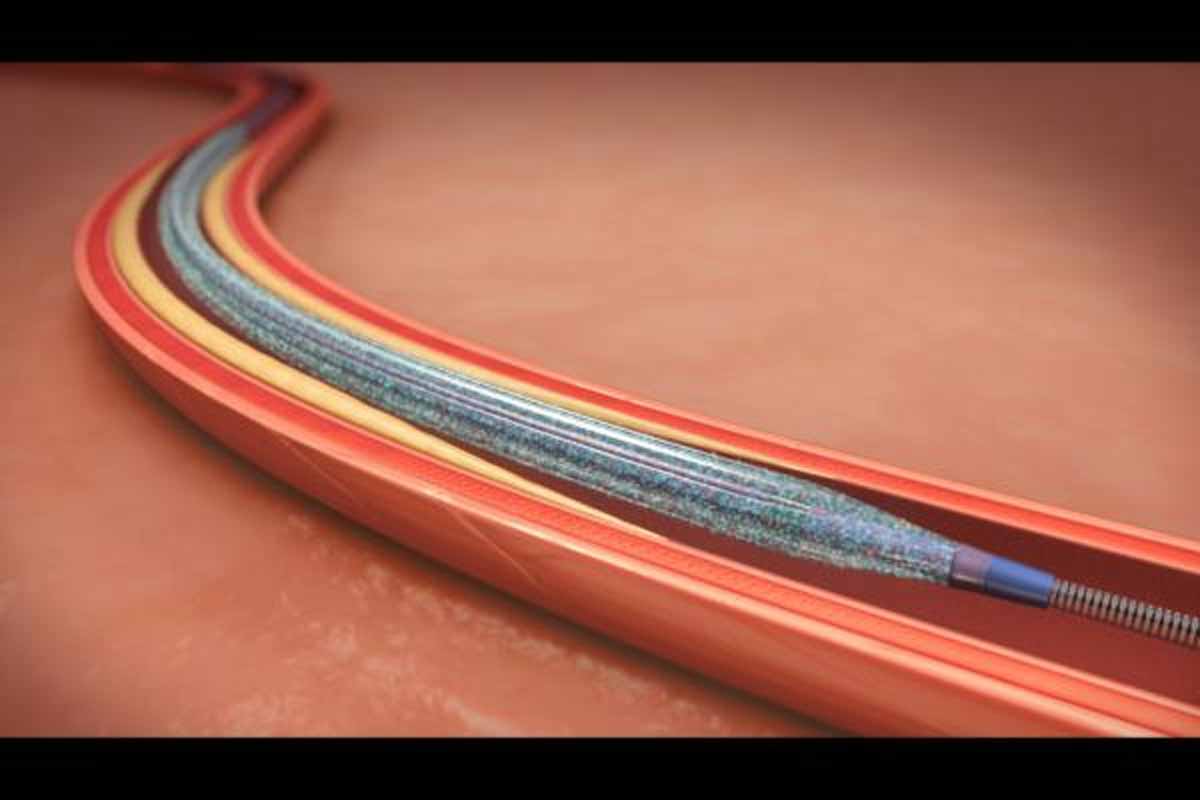
In PCI patients at high risk for bleeding, a polymer-based DES combined with 1-month dual antiplatelet therapy (DAPT) imay confer better angiographic outcomes than a polymer-free drug eluding stent combined with the same lppDAPT regimen finds Onyx ONE trial.The findings were presented at the 31st annual Transcatheter Cardiovascular Therapeutics (TCT) scientific symposium.
Onyx ONE, headed by Stephan Windecker, MD (Swiss Cardiovascular Center, Bern, Switzerland), found that for the primary composite safety endpoint of cardiac death, MI, and definite/probable stent thrombosis at 1 year, the Resolute Onyx zotorolimus-eluting stent (ZES) was noninferior to the drug-coated BioFreedom stent, with a risk difference of 0.2% and a one-sided upper-bound 95% CI of 3%.
Up to 40% of patients undergoing PCI have high bleeding risk (HBR) characteristics and current guidelines recommend three to six months of DAPT for HBR patients undergoing PCI with consideration for DAPT duration as short as one month in selected patients. However, there are scant randomized data with shorter durations of DAPT, especially for current generation polymer-based DES. The Onyx ONE trial compared a polymer-based DES compared to polymer-free drug-coated stents (DCS) in HBR patients treated with one-month DAPT (aspirin and an oral P2Y12 inhibitor).
The prospective, multicenter, single-blind randomized trial enrolled 2,000 HBR patients who were randomized 1:1 at 84 global sites. The primary safety endpoint was cardiac death, myocardial infarction (MI) or definite/probable stent thrombosis at one year. The powered secondary endpoint was target lesion failure, a composite of cardiac death, target vessel myocardial infarction, or clinically driven target lesion revascularization. Other secondary endpoints included device and procedure success rates, BARC bleeding, and the individual components of the primary endpoints.
Of the 1,996 patients randomized, one-year follow-up was available in 98.5% (n=988) of the ZES arm and 97.6% (n=969) of the DCS arm. Baseline and lesion characteristics were similar among both groups. Crossover to an alternative stent occurred more frequently in the DCS arm (40 vs. 2, p<0.001), and ZES showed improved angiographic outcomes resulting in greater device success post-PCI (92.8% for ZES and 89.7% for DCS, p=0.007). At two months after PCI, 92% of patients were on single antiplatelet therapy and 88% were at one year. The primary safety endpoint of cardiac death, MI or definite/probable stent thrombosis at one year was 17.1% for ZES and 16.9% for DCS (Pnon-inferiority = 0.011). In addition, the powered secondary effectiveness endpoint of TLF at one year was 18.0% for ZES and 17.9% for DCS (HR 1.02, 95% CI [0.83, 1.26], p=0.84). Bleeding rates were also similar among both treatment groups.
“The Onyx ONE global randomized trial is the first study to evaluate outcomes between a polymer-based DES and a polymer-free DCS with one-month DAPT after PCI in a complex patient population,” said Stephan Windecker, MD, Director and Chief Physician, Department of Cardiology at the Swiss Cardiovascular Center in Bern, Switzerland. “The data shows that the polymer-based zotarolimus-eluting stent is as safe and effective as the polymer-free drug-coated stent in HBR patients treated with one-month DAPT and provides important evidence to inform clinical decision-making for high bleeding risk patients.”
The Onyx ONE study was funded by Medtronic. Dr. Windecker disclosed research grants to the institution from Abbott, Amgen, Bayer, Bristol Myers-Squibb, Boston Scientific, Biotronik, CLS Behring, Edwards Lifesciences, Medtronic, Polares, and Sinomed.
Dr Prem Aggarwal, (MD, DNB Medicine, DNB Cardiology) is a Cardiologist by profession and also the Co-founder of Medical Dialogues. He is the Chairman of Sanjeevan Hospital in Central Delhi and also serving as the member of Delhi Medical Council

Disclaimer: This site is primarily intended for healthcare professionals. Any content/information on this website does not replace the advice of medical and/or health professionals and should not be construed as medical/diagnostic advice/endorsement or prescription. Use of this site is subject to our terms of use, privacy policy, advertisement policy. © 2020 Minerva Medical Treatment Pvt Ltd