- Home
- Editorial
- News
- Practice Guidelines
- Anesthesiology Guidelines
- Cancer Guidelines
- Cardiac Sciences Guidelines
- Critical Care Guidelines
- Dentistry Guidelines
- Dermatology Guidelines
- Diabetes and Endo Guidelines
- Diagnostics Guidelines
- ENT Guidelines
- Featured Practice Guidelines
- Gastroenterology Guidelines
- Geriatrics Guidelines
- Medicine Guidelines
- Nephrology Guidelines
- Neurosciences Guidelines
- Obs and Gynae Guidelines
- Ophthalmology Guidelines
- Orthopaedics Guidelines
- Paediatrics Guidelines
- Psychiatry Guidelines
- Pulmonology Guidelines
- Radiology Guidelines
- Surgery Guidelines
- Urology Guidelines
Glucose monitoring system not requiring blood sample calibration
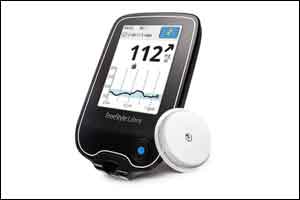
The US Food and Drug Administration (FDA) has approved the FreeStyle Libre Flash Glucose Monitoring System, the first continuous glucose monitoring system that can be used by adult patients to make diabetes treatment decisions without calibration using a blood sample from the fingertip (often referred to as a “fingerstick”). It is intended for use in people 18 years of age and older with diabetes; after a 12-hour start-up period, it can be worn for up to 10 days.
The system reduces the need for fingerstick testing by using a small sensor wire inserted below the skin’s surface that continuously measures and monitors glucose levels. Users can determine glucose levels by waving a dedicated, mobile reader above the sensor wire to determine if glucose levels are too high (hyperglycemia) or too low (hypoglycemia), and how glucose levels are changing. It is intended for use in people 18 years of age and older with diabetes; after a 12-hour start-up period, it can be worn for up to 10 days.
“The FDA is always interested in new technologies that can help make the care of people living with chronic conditions, such as diabetes, easier and more manageable,” said Donald St. Pierre, acting director of the Office of In Vitro Diagnostics and Radiological Health and deputy director of new product evaluation in the FDA’s Center for Devices and Radiological Health. “This system allows people with diabetes to avoid the additional step of fingerstick calibration, which can sometimes be painful, but still provides necessary information for treating their diabetes with a wave of the mobile reader.”
People with diabetes must regularly test and monitor their blood sugar to make sure it is at an appropriate level, which is often done multiple times per day by taking a fingerstick sample and testing it with a blood glucose meter. Typically patients use results of a traditional fingerstick test to make diabetes treatment decisions; however, fingerstick testing is not needed to inform appropriate care choices or to calibrate glucose levels with this system.
According to the Centers for Disease Control and Prevention, more than 29 million people in the U.S. have diabetes. People with diabetes either do not make enough insulin (type 1 diabetes) or cannot use insulin properly (type 2 diabetes). When the body doesn’t have enough insulin or cannot use it effectively, sugar builds up in the blood. High blood sugar levels can lead to heart disease; stroke; blindness; kidney failure; and amputation of toes, feet or legs.
The FDA evaluated data from a clinical study of individuals aged 18 and older with diabetes, and reviewed the device’s performance by comparing readings obtained by the FreeStyle Libre Glucose Monitoring System to those obtained by an established laboratory method used for analysis of blood glucose.
Risks associated with use of the system may include hypoglycemia or hyperglycemia in cases where information provided by the device is inaccurate and used to make treatment decisions, as well as mild skin irritations around the insertion site. It does not provide real-time alerts or alarms in the absence of a user-initiated action; for example, it cannot alert users to low blood glucose levels while they are asleep.
The FreeStyle Libre Flash Glucose Monitoring System is manufactured by Abbott Diabetes Care Inc.

Disclaimer: This site is primarily intended for healthcare professionals. Any content/information on this website does not replace the advice of medical and/or health professionals and should not be construed as medical/diagnostic advice/endorsement or prescription. Use of this site is subject to our terms of use, privacy policy, advertisement policy. © 2020 Minerva Medical Treatment Pvt Ltd