- Home
- Editorial
- News
- Practice Guidelines
- Anesthesiology Guidelines
- Cancer Guidelines
- Cardiac Sciences Guidelines
- Critical Care Guidelines
- Dentistry Guidelines
- Dermatology Guidelines
- Diabetes and Endo Guidelines
- Diagnostics Guidelines
- ENT Guidelines
- Featured Practice Guidelines
- Gastroenterology Guidelines
- Geriatrics Guidelines
- Medicine Guidelines
- Nephrology Guidelines
- Neurosciences Guidelines
- Obs and Gynae Guidelines
- Ophthalmology Guidelines
- Orthopaedics Guidelines
- Paediatrics Guidelines
- Psychiatry Guidelines
- Pulmonology Guidelines
- Radiology Guidelines
- Surgery Guidelines
- Urology Guidelines
Esophageal cancer-Standard Treatment Guidelines

Ministry of Health and Family Welfare, Government of India has issued the Standard Treatment Guidelines for Esophageal cancer.
Following are the major recommendations :
WHEN TO SUSPECT
Presenting Symptoms
Most common presenting feature of esophageal cancer is dysphagia, initially to solids and then to liquids. Other symptoms that patient may present with are
Investigations
Diagnostic Investigations
1. Barium Swallow (optional) : This continues to be the first investigation in majority of patients presenting with dysphagia. It gives information regarding the 1. Site 2. length of lesion 3. Morphology (Proliferative/Stricturous/ulcerative or combination) 4. Extra esophageal spread (axis deviation, sinuses and fistulation).
2. Esophagoscopy : Fiberoptic esophagoscopy is essential for biopsy/cytology.
Staging Investigations
1. CT scan : Chest and upper abdomen
2. Endoscopic ultrasonography (EUS)
3. Fiberoptic bronchoscopy : for tumours located at and above the level of the carina.
CT scan and EUS are complimentary for assessing the lateral extension of disease and lymph node status. EUS scores over CT scan in assessment of the depth of tumour invasion, particularly in early cancer, and status of regional lymph node. However, in stricturous lesions, EUS may not always be possible. CT scan is equally accurate in assessment of T3/T4 lesions; abdominal CT scan additionally can screen liver and coeliac lymph nodes. Bronchoscopy is an essential non invasive investigation for assessing the tracheo-bronchial tree for early or frank invasion. It is recommended prior to surgery or radiation for upper and mid esophageal disease.
PET scan can effectively detect presence of disseminated disease. However, presently it is an investigational modality of investigation. Thoracoscopy and laparoscopy for staging has been investigated and reported increased rate of detecting positive lymph nodes than non invasive staging modalities (Level IIb).
Routine Investigations for assessing fitness for treatment
Treatment Options
Two factors determine the treatment:
1. General condition or the Performance status
2. Stage
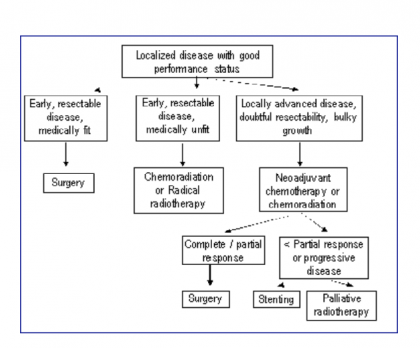
Stage 0 (TisN0M0)
Patients are rarely diagnosed in this stage. The treatment of choice is surgery. If the disease is localised (preferably T1a), Endoscopic mucosal resection (EMR) can be offered in centres with expertise provided the patient is reliable for followup. For more extensive disease, esophagectomy is the treatment of choice.
Stage I (T1N0M0)
Surgery is the treatment of choice. Radiation therapy may be offered if the patient is medically unfit or not willing for surgery.
Stage II/III (T2N0M0, T3N0M0, T1N1M0, T2N1M0)
Surgery for T2 and T3 lesions
Surgery for T4 lesions with limited infiltration of pleura or pericardium which is amenable to complete resection.
Neo adjuvant chemotherapy/concomitant chemo-radiation for T3/T4 tumours which are bulky or of doubtful resectability. If there is complete or partial response to neo adjuvant therapy and tumour appears resectable patient should proceed for surgery; if the response is sub optimal and disease appears non resectable, patient should either proceed for radiation or palliative therapy (see below).
Investigational treatments
1. Chemoradiotherapy alone or chemoradiotherapy followed by surgery.
2. Neo adjuvant Chemotherapy followed by surgery.
3. Post operative radiation therapy.
Stage IVa (ant T, any N, M1a)
Surgery (per primum or following neo adjuvant therapy): If the disease is operable in the absence of distant metastases. However, more than 50% of patients will have distant metastases. Such patients will be candidates for palliative treatment.
Principles of Surgery
Surgical Approach
Adenocarcinoma of the cardio-esophageal junction can be resected through a left thoracoabdominal approach. Surgery involves mobilization of the oesophagus upto the inferior pulmonary vein along with dissection of lower paraesophageal lymph nodes, standard mobilization of stomach along with D2 lymphadenectomy. Gastro-oesophageal anastomoses could be either mechanical (using stapler) or hand sewn.
Adenocarcinoma of the distal portion of the oesophagus or cardio-esophageal junction extending into the lower oesophagus where the proximal extent of the tumour is such that adequate margin is not possible through the left thoraco-abdominal approach should be treated by either trans thoracic or trans hiatal esophagectomy. Phase III trial comparing transhiatal esophagectomy to transthoracic esophagectomy and lymphadenectomy for adenocarcinoma of the oesophagus did not find difference in median overall and disease free survival between the two procedures. However, there was a trend towards superior long term (5-year) survival, not reaching statistical significance, in favour of transthoracic esophagectomy (Level Ib).
Carcinoma of the lower, mid and upper esophagus (excluding cervical oesophagus) is managed either by transhiatal or trans thoracic esophagectomy and esophago-gastric anastomosis in the neck or thorax. There is no consensus as to the best or the ideal surgical approach.
Transthoracic esophagectomy has the advantage of mobilization of the oesophagus under vision. Also, systematic mediastinal lymph node dissection can be performed. Trans hiatal approach, according to proponents, is less morbid with fewer pulmonary complications. There are four published Phase three trials comparing the two approaches. Three of these had few patients and thus, meaningful conclusions cannot be drawn. The fourth and the latest trial has 220 patients, all adenocarcinoma restricted to the distal oesophagus or cardio-esophageal junction. There was no difference in the median overall survival; however, there was a trend towards a survival benefit at five years with the trans thoracic approach (Level Ib). The published meta analysis of over 60 trials (both prospective and retrospective) comparing transhiatal to transthoracic esophagectomy did not find any difference in the overall survival (Level IIc). Till results of large randomized trials are available the preferred surgical approach will continue to be biased by surgeons’ choice.
Extent of lymphadenectomy
Lymph node metastasis is one of the most important prognostic factors for carcinoma of the oesophagus. Since the oesophagus has extensive lymphatic network and most patients present with advanced disease, the majority of patients undergoing surgery have lymph node metastases. Three field lymph node dissection (lower cervical, mediastinal and abdominal) is reported to improve survival without increased procedure related morbidity and mortality (Level IIa). However, most reported studies are small or have compared results with historical controls. The only one randomized trial of over 60 patients has reported higher, though not statistically significant, survival in patients undergoing three field lymph node dissections (Level Ib). Extensive lymph node dissection provides ‘accurate nodal staging’ resulting in stage migration and apparent ‘improvement in survival’. In absence of conclusive Level I evidence, the advantage of three field lymph node dissection over the conventional limited lymph node dissection remains speculative. In fact, an adequately powered randomized trial could answer the question regarding the importance of lymph node dissection in management of carcinoma oesophagus and indirectly address the issue of transhiatal versus transthoracic approach.
Definitive radiation and chemo-radiation therapy
Two published (RTOG and ECOG) randomized trials have reported better overall survival with concomitant chemo-radiation than radiation therapy alone. However, increasing the dose of radiation therapy (50.4 versus 64.8) in concomitant setting did not result in increased survival (Inter Group trial). Meta analysis of 13 trials combining radiation with chemotherapy published in the Cochrane library has reported an absolute reduction in the mortality and local recurrence rate of 7% and 12% respectively in favour of combination therapy. The combination treatment is associated with increased life threatening toxicities (Level Ia). There are no trials comparing concomitant chemo-radiation with surgery alone. However two trials comparing surgery to radiation alone have reported better survival with surgery (Level Ib). Hence based on the available evidence, if a patient is to be treated with definitive radiation, it should be combined with chemotherapy, provided performance status is optimal.
Surgery as adjuvant to radiation, chemotherapy or combination of both
Pre-operative radiotherapy
A meta analysis as well as the five published randomized trials comparing preoperative radiation therapy to surgery alone have not shown benefit of pre operative radiation over surgery alone (Level Ia).
Pre-operative concomitant chemo-radiation
There are eleven major trials and three meta analysis comparing preoperative concomitant chemoradiation to surgery alone. Meta analysis of pre operative chemoradiation and surgery to surgery alone has reported improved survival and reduced loco-regional recurrence (Level Ia). However, combination treatment is associated with trend towards increased treatment related morbidity and mortality. In the absence of results from a large trial and increased treatment related morbidity, neo adjuvant chemo-radiation should be considered as one of the options of treating localized disease.
Pre-operative chemotherapy
There are nine major published trials of pre-operative chemotherapy in the management of carcinoma of the oesophagus. The two large trials have reported results which are divergent. The Intergroup trial of 440 patients reported by Kelsen et al observed no improvement in survival with pre operative combination of cisplatin and fluorouracil among patients with adenocarcinoma or epidermoid carcinoma of the oesophagus. The MRC trial of 802 patients reported improved survival with two cycles of cisplatin and fluorouracil without additional serious events. The meta analysis of all trials put together concludes that preoperative chemotherapy plus surgery appears to offer a survival advantage at 3, 4, and 5 years, which reached significance only at 5 years compared to surgery alone for resectable thoracic esophageal cancer of any histologic type. The number needed to treat for one extra survivor at five years is eleven patients. (Level Ia).
Post-operative radiotherapy
Three trials have compared surgery and post operative radiation to surgery alone. The Chinese trial of 495 patients observed improved 5-year survival in patients with positive lymph nodes and stage III disease receiving post operative radiation. However, the difference in the overall survival between the two groups was statistically not different. The meta analysis of all three trials also does not show benefit of post operative radiotherapy. Therefore, in the absence of Level I evidence post operative radiotherapy is indicated only for patients with positive margin and residual disease.
Post operative chemotherapy
Phase III trials of surgery and post operative chemotherapy have not reported survival benefit over surgery alone. A Phase III study by Japanese Clinical Oncology Group (JCOG) reported better disease free survival at 5-year with post operative chemotherapy; however there was no difference in the overall survival (Level Ia).In adenocarcinoma of the cardio oesophageal junction (and stomach) two trials – the MAGIC trial evaluating perioperative chemotherapy with three cycles of ECF given pre as well as 3 cycles given postoperatively, and the MacDonald post operative chemo-radiotherapy trial – both showed improved overall survival (Level Ib). Thus in patients with adenocarcinoma of the cardia having good performance status, perioperative chemotherapy (3 preoperative cycles of ECF and 3 postoperative cycles of ECF) or post operative chemo-radiation should be the standard of care.
Principles of Radiation therapy
Radical radiotherapy
The inclusion criteria are :
- All lesions (except stenotic) in upper / mid / lower esophagus
- Lesion £ than 5 cm on barium swallow and esophagoscopy
- Histologically proven esophageal carcinoma
- Karnofsky Performance Status (KPS) of > 60%
- Age £ 60 years.
- Metastatic work - up negative (No palpable S/C nodes, Bronchoscopy & USG abdomen normal).
Dose : 60 - 64.8Gy / 33 - 36 fractions, with reducing fields.
Portal design : Extended field: esophageal lesion including the lymph drainage areas, with 5 cm margin on either side upto 39.6Gy / 22 fractions/ 4.5 weeks.
Reduced fields/ boost: Lesion with 2 - 3 cm. margins, with oblique portals, upto 60 - 64.8Gy / 33 - 36 fractions.
When feasible, external Radiotherapy can be combined with Intraluminal radiotherapy (ILRT) as a boost.
Dose of EBRT : 50.4Gy / 28 fractions with reducing fields.
ILRT Boost : 5 - 8Gy / 2-3 fractions high dose rate (HDR), one week apart or single fraction 20Gy low dose rate (LDR).
Concomitant chemo-radiation regimen
Principles of Chemotherapy
Neoadjuvant chemotherapy protocols
Palliative treatment
If the general condition is good,
Palliative radiotherapy
The intent of treatment is to achieve quick and good palliation in the form of relief of dysphagia and pain.
The inclusion criteria are :
- Lesions in upper / mid / lower esophagus
- Lesion £ 10 cm long on barium swallow and esophagoscopy
- Histologically proven esophageal carcinoma
- Karnofsky performance status (KPS) of £ 50%
- Recurrent / metastatic disease.
Dose : 3000cGy /10 fractions /2 weeks
Portal : Esophageal lesion with 2-3 cm margin
Evaluation and response assessment is done after 4 - 6 weeks and further external Radiotherapy or Brachytherapy boost may be delivered.
Reduced field / boost : 2000cGy/10# / 2 weeks, using oblique portals
Palliative radiation can also be delivered in the form of ILRT alone or in combination with EBRT. The dose per fraction ranges from 5 - 8Gy, in 2- 3 fractions, one week apart. There is no difference in local control or survival between high dose rate brachytherapy compared with external beam radiation. (Level II)
Investigational Treatment
Palliative chemotherapy with intubation if associated with significant dysphagia. Response rates ranging from 30% to 50% and one year survival ranging from 0% to 5% is reported with platinum based combination chemotherapy.
If the general condition is poor with limited life expectancy
Treatment of esophageal fistula
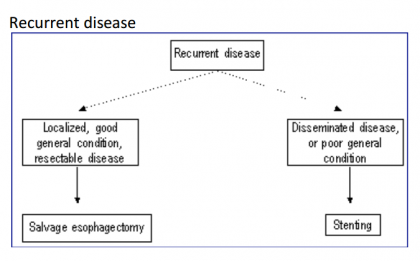
Treatment of Recurrent disease
1. Salvage surgery for localised resectable failures.
2. Palliative treatment or supportive care alone as described before.
Management algorithm for esophageal cancer
Workup
1. Barium swallow (optional)
2. Upper GI scopy with biopsy / cytology
3. CT scan chest and upper abdomen
4. Fiber optic bronchoscopy – for upper and middle third growths
5. Routine hematology, biochemistry
6. Pulmonary function tests – if surgery contemplated
a. Assess stage of disease – (i) Localized (ii) Disseminated
b. Assess performance and nutritional status
Disseminated disease with good performance status
Poor performance status and / or nutritional status
Best supportive care
Dr Anil K. D’ Cruz
Director and
Chief Head and Neck Services,
Tata Memorial Hospital, Mumbai
Following are the major recommendations :
WHEN TO SUSPECT
Presenting Symptoms
Most common presenting feature of esophageal cancer is dysphagia, initially to solids and then to liquids. Other symptoms that patient may present with are
- Odynophagia
- Dyspepsia
- Loss of weight
- Anorexia
- Left supraclavicular node
- Haematemesis
- Vomiting
Investigations
Diagnostic Investigations
1. Barium Swallow (optional) : This continues to be the first investigation in majority of patients presenting with dysphagia. It gives information regarding the 1. Site 2. length of lesion 3. Morphology (Proliferative/Stricturous/ulcerative or combination) 4. Extra esophageal spread (axis deviation, sinuses and fistulation).
2. Esophagoscopy : Fiberoptic esophagoscopy is essential for biopsy/cytology.
Staging Investigations
1. CT scan : Chest and upper abdomen
2. Endoscopic ultrasonography (EUS)
3. Fiberoptic bronchoscopy : for tumours located at and above the level of the carina.
CT scan and EUS are complimentary for assessing the lateral extension of disease and lymph node status. EUS scores over CT scan in assessment of the depth of tumour invasion, particularly in early cancer, and status of regional lymph node. However, in stricturous lesions, EUS may not always be possible. CT scan is equally accurate in assessment of T3/T4 lesions; abdominal CT scan additionally can screen liver and coeliac lymph nodes. Bronchoscopy is an essential non invasive investigation for assessing the tracheo-bronchial tree for early or frank invasion. It is recommended prior to surgery or radiation for upper and mid esophageal disease.
PET scan can effectively detect presence of disseminated disease. However, presently it is an investigational modality of investigation. Thoracoscopy and laparoscopy for staging has been investigated and reported increased rate of detecting positive lymph nodes than non invasive staging modalities (Level IIb).
Routine Investigations for assessing fitness for treatment
- Hemogram
- Liver Function Test/Renal Function Test
- Chest X-ray
- Pulmonary Function Test
- ECG
Treatment Options
Two factors determine the treatment:
1. General condition or the Performance status
2. Stage
- General condition or performance status is an important factor in determining the treatment of a patient with cancer oesophagus. Dysphagia, particularly if it is long standing and complete, leads to chronic dehydration and malnutrition. Such patients will not tolerate surgery, radiation or chemotherapy. Supportive care to optimise general condition should be the priority. Subsequently, if performance status improves, definitive treatment can be contemplated depending on stage of the disease.
- Stage : Patients with localized disease are ideally treated with surgery in the absence of medical contraindications. As per the staging, presence of abdominal or celiac lymph nodes is classified as disseminated disease. However prognosis of patients with abdominal or celiac lymph node metastasis is not the same as that with systemic distant metastasis. Hence, patients with operable local disease should be offered surgery with appropriate lymphadenectomy.
- The preferred treatment of carcinoma of the cervical oesophagus is radical radiotherapy or concomitant chemoradiotherapy.
Stage 0 (TisN0M0)
Patients are rarely diagnosed in this stage. The treatment of choice is surgery. If the disease is localised (preferably T1a), Endoscopic mucosal resection (EMR) can be offered in centres with expertise provided the patient is reliable for followup. For more extensive disease, esophagectomy is the treatment of choice.
Stage I (T1N0M0)
Surgery is the treatment of choice. Radiation therapy may be offered if the patient is medically unfit or not willing for surgery.
Stage II/III (T2N0M0, T3N0M0, T1N1M0, T2N1M0)
Surgery for T2 and T3 lesions
Surgery for T4 lesions with limited infiltration of pleura or pericardium which is amenable to complete resection.
Neo adjuvant chemotherapy/concomitant chemo-radiation for T3/T4 tumours which are bulky or of doubtful resectability. If there is complete or partial response to neo adjuvant therapy and tumour appears resectable patient should proceed for surgery; if the response is sub optimal and disease appears non resectable, patient should either proceed for radiation or palliative therapy (see below).
Investigational treatments
1. Chemoradiotherapy alone or chemoradiotherapy followed by surgery.
2. Neo adjuvant Chemotherapy followed by surgery.
3. Post operative radiation therapy.
Stage IVa (ant T, any N, M1a)
Surgery (per primum or following neo adjuvant therapy): If the disease is operable in the absence of distant metastases. However, more than 50% of patients will have distant metastases. Such patients will be candidates for palliative treatment.
Principles of Surgery
Surgical Approach
- Esophago-gastrectomy through left thoraco-abdominal approach (Garlock procedure) : for adenocarcinoma of the cardio-esophageal junction.
- Trans thoracic esophagectomy with intrathoracic anastomosis (Ivor Lewis procedure).
- Trans thoracic total esophagectomy with cervical anastomosis.
- Trans hiatal esophagectomy with cervical anastomosis.
Adenocarcinoma of the cardio-esophageal junction can be resected through a left thoracoabdominal approach. Surgery involves mobilization of the oesophagus upto the inferior pulmonary vein along with dissection of lower paraesophageal lymph nodes, standard mobilization of stomach along with D2 lymphadenectomy. Gastro-oesophageal anastomoses could be either mechanical (using stapler) or hand sewn.
Adenocarcinoma of the distal portion of the oesophagus or cardio-esophageal junction extending into the lower oesophagus where the proximal extent of the tumour is such that adequate margin is not possible through the left thoraco-abdominal approach should be treated by either trans thoracic or trans hiatal esophagectomy. Phase III trial comparing transhiatal esophagectomy to transthoracic esophagectomy and lymphadenectomy for adenocarcinoma of the oesophagus did not find difference in median overall and disease free survival between the two procedures. However, there was a trend towards superior long term (5-year) survival, not reaching statistical significance, in favour of transthoracic esophagectomy (Level Ib).
Carcinoma of the lower, mid and upper esophagus (excluding cervical oesophagus) is managed either by transhiatal or trans thoracic esophagectomy and esophago-gastric anastomosis in the neck or thorax. There is no consensus as to the best or the ideal surgical approach.
Transthoracic esophagectomy has the advantage of mobilization of the oesophagus under vision. Also, systematic mediastinal lymph node dissection can be performed. Trans hiatal approach, according to proponents, is less morbid with fewer pulmonary complications. There are four published Phase three trials comparing the two approaches. Three of these had few patients and thus, meaningful conclusions cannot be drawn. The fourth and the latest trial has 220 patients, all adenocarcinoma restricted to the distal oesophagus or cardio-esophageal junction. There was no difference in the median overall survival; however, there was a trend towards a survival benefit at five years with the trans thoracic approach (Level Ib). The published meta analysis of over 60 trials (both prospective and retrospective) comparing transhiatal to transthoracic esophagectomy did not find any difference in the overall survival (Level IIc). Till results of large randomized trials are available the preferred surgical approach will continue to be biased by surgeons’ choice.
Extent of lymphadenectomy
Lymph node metastasis is one of the most important prognostic factors for carcinoma of the oesophagus. Since the oesophagus has extensive lymphatic network and most patients present with advanced disease, the majority of patients undergoing surgery have lymph node metastases. Three field lymph node dissection (lower cervical, mediastinal and abdominal) is reported to improve survival without increased procedure related morbidity and mortality (Level IIa). However, most reported studies are small or have compared results with historical controls. The only one randomized trial of over 60 patients has reported higher, though not statistically significant, survival in patients undergoing three field lymph node dissections (Level Ib). Extensive lymph node dissection provides ‘accurate nodal staging’ resulting in stage migration and apparent ‘improvement in survival’. In absence of conclusive Level I evidence, the advantage of three field lymph node dissection over the conventional limited lymph node dissection remains speculative. In fact, an adequately powered randomized trial could answer the question regarding the importance of lymph node dissection in management of carcinoma oesophagus and indirectly address the issue of transhiatal versus transthoracic approach.
Definitive radiation and chemo-radiation therapy
Two published (RTOG and ECOG) randomized trials have reported better overall survival with concomitant chemo-radiation than radiation therapy alone. However, increasing the dose of radiation therapy (50.4 versus 64.8) in concomitant setting did not result in increased survival (Inter Group trial). Meta analysis of 13 trials combining radiation with chemotherapy published in the Cochrane library has reported an absolute reduction in the mortality and local recurrence rate of 7% and 12% respectively in favour of combination therapy. The combination treatment is associated with increased life threatening toxicities (Level Ia). There are no trials comparing concomitant chemo-radiation with surgery alone. However two trials comparing surgery to radiation alone have reported better survival with surgery (Level Ib). Hence based on the available evidence, if a patient is to be treated with definitive radiation, it should be combined with chemotherapy, provided performance status is optimal.
Surgery as adjuvant to radiation, chemotherapy or combination of both
Pre-operative radiotherapy
A meta analysis as well as the five published randomized trials comparing preoperative radiation therapy to surgery alone have not shown benefit of pre operative radiation over surgery alone (Level Ia).
Pre-operative concomitant chemo-radiation
There are eleven major trials and three meta analysis comparing preoperative concomitant chemoradiation to surgery alone. Meta analysis of pre operative chemoradiation and surgery to surgery alone has reported improved survival and reduced loco-regional recurrence (Level Ia). However, combination treatment is associated with trend towards increased treatment related morbidity and mortality. In the absence of results from a large trial and increased treatment related morbidity, neo adjuvant chemo-radiation should be considered as one of the options of treating localized disease.
Pre-operative chemotherapy
There are nine major published trials of pre-operative chemotherapy in the management of carcinoma of the oesophagus. The two large trials have reported results which are divergent. The Intergroup trial of 440 patients reported by Kelsen et al observed no improvement in survival with pre operative combination of cisplatin and fluorouracil among patients with adenocarcinoma or epidermoid carcinoma of the oesophagus. The MRC trial of 802 patients reported improved survival with two cycles of cisplatin and fluorouracil without additional serious events. The meta analysis of all trials put together concludes that preoperative chemotherapy plus surgery appears to offer a survival advantage at 3, 4, and 5 years, which reached significance only at 5 years compared to surgery alone for resectable thoracic esophageal cancer of any histologic type. The number needed to treat for one extra survivor at five years is eleven patients. (Level Ia).
Post-operative radiotherapy
Three trials have compared surgery and post operative radiation to surgery alone. The Chinese trial of 495 patients observed improved 5-year survival in patients with positive lymph nodes and stage III disease receiving post operative radiation. However, the difference in the overall survival between the two groups was statistically not different. The meta analysis of all three trials also does not show benefit of post operative radiotherapy. Therefore, in the absence of Level I evidence post operative radiotherapy is indicated only for patients with positive margin and residual disease.
Post operative chemotherapy
Phase III trials of surgery and post operative chemotherapy have not reported survival benefit over surgery alone. A Phase III study by Japanese Clinical Oncology Group (JCOG) reported better disease free survival at 5-year with post operative chemotherapy; however there was no difference in the overall survival (Level Ia).In adenocarcinoma of the cardio oesophageal junction (and stomach) two trials – the MAGIC trial evaluating perioperative chemotherapy with three cycles of ECF given pre as well as 3 cycles given postoperatively, and the MacDonald post operative chemo-radiotherapy trial – both showed improved overall survival (Level Ib). Thus in patients with adenocarcinoma of the cardia having good performance status, perioperative chemotherapy (3 preoperative cycles of ECF and 3 postoperative cycles of ECF) or post operative chemo-radiation should be the standard of care.
Principles of Radiation therapy
Radical radiotherapy
The inclusion criteria are :
- All lesions (except stenotic) in upper / mid / lower esophagus
- Lesion £ than 5 cm on barium swallow and esophagoscopy
- Histologically proven esophageal carcinoma
- Karnofsky Performance Status (KPS) of > 60%
- Age £ 60 years.
- Metastatic work - up negative (No palpable S/C nodes, Bronchoscopy & USG abdomen normal).
- External beam radiotherapy (EBRT) alone
Dose : 60 - 64.8Gy / 33 - 36 fractions, with reducing fields.
Portal design : Extended field: esophageal lesion including the lymph drainage areas, with 5 cm margin on either side upto 39.6Gy / 22 fractions/ 4.5 weeks.
Reduced fields/ boost: Lesion with 2 - 3 cm. margins, with oblique portals, upto 60 - 64.8Gy / 33 - 36 fractions.
- External beam radiotherapy and brachytherapy
When feasible, external Radiotherapy can be combined with Intraluminal radiotherapy (ILRT) as a boost.
Dose of EBRT : 50.4Gy / 28 fractions with reducing fields.
ILRT Boost : 5 - 8Gy / 2-3 fractions high dose rate (HDR), one week apart or single fraction 20Gy low dose rate (LDR).
Concomitant chemo-radiation regimen
- 50Gy in 25 fractions over 5 weeks, plus cisplatin intravenously on the first day of weeks 1, 5, 8, and 11, and fluorouracil, 1g/m2 per day by continuous infusion on the first 4 days of weeks 1, 5, 8, and 11. (RTOG regimen).
- 60Gy in 6 to 61/2 weeks. Chemotherapy to be initiated within 24 hours after the commencement of radiation therapy. 5-FU to be delivered by continuous infusion for 96 hours starting on day 2 at the rate of 1000 mg per m2 over 24 hours. This regimen of 5- FU to be repeated once again beginning on day 28. Bolus injection of mitomycin C (10 mg per m2) to be administered on day 2 and not to be repeated. The dose of mitomycin C not to exceed 18 mg and the dose of 5-FU not to exceed 1800 mg over a 24-hour period. (ECOG regimen).
Principles of Chemotherapy
Neoadjuvant chemotherapy protocols
- Two 4-day cycles, 3 weeks apart, of cisplatin 80 mg/m(2) by infusion over 4 h plus fluorouracil 1000 mg/m(2) daily by continuous infusion for 4 days. (MRC protocol).
- Cisplatin, at a dose of 100 mg per square meter of body-surface area, given as a rapid intravenous infusion after prehydration on day 1. Immediately thereafter, fluorouracil administered at a dose of 1000 mg per square meter as a continuous infusion from day 1 through day 5 (120 hours) of each cycle. The cycle to be repeated beginning on days 29 and 58. Surgery performed two to four weeks after chemotherapy (Intergroup protocol).
Palliative treatment
If the general condition is good,
- Relief of dysphagia by placement of esophageal stent alone, preferably self expanding metallic stent as these are easy to deploy.
- Radiation therapy with intubation if associated with significant dysphagia.
- Intraluminal radiation therapy alone.
- Endoscopic laser destruction of tumour or electrocoagulation.
Palliative radiotherapy
The intent of treatment is to achieve quick and good palliation in the form of relief of dysphagia and pain.
The inclusion criteria are :
- Lesions in upper / mid / lower esophagus
- Lesion £ 10 cm long on barium swallow and esophagoscopy
- Histologically proven esophageal carcinoma
- Karnofsky performance status (KPS) of £ 50%
- Recurrent / metastatic disease.
Dose : 3000cGy /10 fractions /2 weeks
Portal : Esophageal lesion with 2-3 cm margin
Evaluation and response assessment is done after 4 - 6 weeks and further external Radiotherapy or Brachytherapy boost may be delivered.
Reduced field / boost : 2000cGy/10# / 2 weeks, using oblique portals
Palliative radiation can also be delivered in the form of ILRT alone or in combination with EBRT. The dose per fraction ranges from 5 - 8Gy, in 2- 3 fractions, one week apart. There is no difference in local control or survival between high dose rate brachytherapy compared with external beam radiation. (Level II)
Investigational Treatment
Palliative chemotherapy with intubation if associated with significant dysphagia. Response rates ranging from 30% to 50% and one year survival ranging from 0% to 5% is reported with platinum based combination chemotherapy.
If the general condition is poor with limited life expectancy
- Nasogastric tube placement for feeding if possible.
- Supportive care.
Treatment of esophageal fistula
- Esophageal intubation with stent.
- Oesophageal and tracheal/bronchial stent placement (double stenting) when possible if the fistula is large or if the tracheal lumen is compromised.
Treatment of Recurrent disease
1. Salvage surgery for localised resectable failures.
2. Palliative treatment or supportive care alone as described before.
Management algorithm for esophageal cancer
Workup
1. Barium swallow (optional)
2. Upper GI scopy with biopsy / cytology
3. CT scan chest and upper abdomen
4. Fiber optic bronchoscopy – for upper and middle third growths
5. Routine hematology, biochemistry
6. Pulmonary function tests – if surgery contemplated
a. Assess stage of disease – (i) Localized (ii) Disseminated
b. Assess performance and nutritional status

Disseminated disease with good performance status
- Stenting
- Palliative chemotherapy
Poor performance status and / or nutritional status
- Stenting

Best supportive care
- Stenting – to relieve obstruction
- Enteral feeding through nasogastric tube or gastrostomy
- Pain relief
Guidelines by The Ministry of Health and Family Welfare :
Dr Anil K. D’ Cruz
Director and
Chief Head and Neck Services,
Tata Memorial Hospital, Mumbai
Next Story
NO DATA FOUND

Disclaimer: This site is primarily intended for healthcare professionals. Any content/information on this website does not replace the advice of medical and/or health professionals and should not be construed as medical/diagnostic advice/endorsement or prescription. Use of this site is subject to our terms of use, privacy policy, advertisement policy. © 2020 Minerva Medical Treatment Pvt Ltd