- Home
- Editorial
- News
- Practice Guidelines
- Anesthesiology Guidelines
- Cancer Guidelines
- Cardiac Sciences Guidelines
- Critical Care Guidelines
- Dentistry Guidelines
- Dermatology Guidelines
- Diabetes and Endo Guidelines
- Diagnostics Guidelines
- ENT Guidelines
- Featured Practice Guidelines
- Gastroenterology Guidelines
- Geriatrics Guidelines
- Medicine Guidelines
- Nephrology Guidelines
- Neurosciences Guidelines
- Obs and Gynae Guidelines
- Ophthalmology Guidelines
- Orthopaedics Guidelines
- Paediatrics Guidelines
- Psychiatry Guidelines
- Pulmonology Guidelines
- Radiology Guidelines
- Surgery Guidelines
- Urology Guidelines
Chronic Cancer Pain Management Guidelines
Cancer pain may not just be from the physical effect of the cancer on a region of the body, but also due to chemicals that the cancer may release in the region of the tumor. Treatment of the cancer can help the pain in these situations.
These guidelines are meant for management of patients with the following pain scenarios:
1)Patients reporting with moderate to severe pain prior to any cancer therapy
2) Patients with advanced disease (including residual or recurrent disease)
3) Patients requiring pain relief during the cancer therapy esp. during chemotherapy or radiotherapy
4) Patient at end of life
5) Pain in cancer survivors
ETIOPATHOLOGY OF PAIN
- Pain can be somatic or visceral or neuropathic in origin. It can either be caused by the disease itself or by cancer therapy or may be unrelated to cancer.
- The pain assessment is largely multidimensional.
- It includes
- The physiological component (nociception);
- The sensory component (i.e. the subject's perception of nociception, such as pain location, intensity and quality).
- The affective component (i.e. mood & anxiety response)
- Cognitive component (i.e. how the pain affects patients' thought processes and how the patients view themselves),
- Behavioral components (i.e. pain behaviors such as analgesic intake or activity level) are assessed during a comprehensive patient assessment.
Cancer Pain Assessment Protocol:
Aim:
To lay down guidelines for
- Comprehensive assessment of patients with cancer for all types of pain
- Appropriate treatment regimens based on the underlying mechanisms causing pain.
Scope of these guidelines:
- Help clinicians assess and manage cancer pain effectively
- To minimize risk of side effects due to analgesics
- To standardize prescriptions and procedures
- To individualize treatment with the goal of maximizing function & quality of life
Assessment of Pain
Who should assess patient's pain?
The patient himself/herself is the most reliable assessor of pain and should, where possible, be the prime assessor of his or her pain. The patient should be the prime assessor of his or her pain. Due to frailty, cognitive impairment or communication deficits not all patients are able to relate the story of their pain. Completion of pain scoring tools may not be possible. In these cases families or health professionals may act as a surrogate.
How should pain be assessed?
Careful history taking and listening attentively to the patient will usually diagnose the type of pain and this in turn dictates the therapy. The severity of the pain will determine the step of the World Health Organization (WHO) ladder at which the therapeutic intervention will begin.
Detailed history of pain:
- Location of pain
- Number of pains
- Intensity of pain : based on patient's self report using;
- 0-10 numerical rating scale or
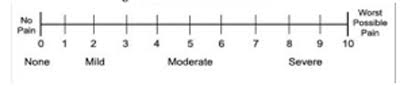
- pictorial scale ( Faces Pain rating scale)
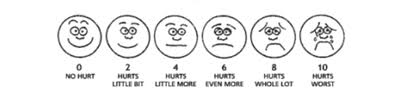
5.Timing of pain: onset, duration, course, intermittent or persistent
6.Characteristics of the pain (Pathophysiology):
- Nociceptive pain: injury to somatic & visceral structures with activation of nociceptors
- Somatic: sharp, well localized, throbbing, aching, stabbing, pressure-like
- Visceral: diffuse, aching, cramping
- Neuropathic pain: injury to peripheral or central nervous system. Described as burning, sharp, shooting, and tingling.
7.Aggravating & relieving factors
8.Interference with daily activities: general activity, sleep, mood, appetite
9.Etiology of pain: Is the pain caused by;
- Cancer
- Cancer therapy: Radiation, chemotherapy, surgery or procedure related pain
- Unrelated cause
7.Analgesic drug history: Current/past pain medication
What medications?
What was the response?
- Pain relief
- Side effects
- Reason for discontinuation
8.Presence of clinically significant psychological disorder , anxiety or depression
9.Oncologic treatment: includes
- Chemotherapy:ongoing & prior
- Radiation therapy
- Surgery
10.Medical history
- Presence of other illnesses/ co-morbidities
- Pre-existing chronic pain
11.Physical examination: Includes general condition, gait of patient, local findings like swelling and inflammation, altered sensation like allodynia, hyperalgesia.
12.Laboratory & imaging studies to evaluate disease progression:
- All available investigations should be reviewed
- In advance cases repeating investigations for new onset pain is not warranted
- However in select cases if the pain presentation is out of proportion of the clinical scenario, relevant imaging should be asked for
13.Reassessment at subsequent follow ups
Pain assessment tools:
Patients with cancer pain should have treatment outcomes monitored regularly using numerical rating scales. Observational pain rating scales should be used in patients who are unable to use a self assessment scale.
The endpoint of assessment is;
- To establish a "Pain Diagnosis": which includes the etiology and pathophysiology of pain
- To Individualize treatment
Use valid pain assessment tools to evaluate, at regular intervals, both pain intensity and the effectiveness of the pain management plan; document these reassessments.
Frequency of assessments:
Continual reassessment of the patient is required to identify the effectiveness of pain management, to identify and treat adverse side effects. All patients should be reassessed after 5-7 days of first visit, when possible.
Guidelines for prescribing Non-opioid analgesics:
- Paracetamol , NSAIDs may be used at any stage of WHO ladder
- Used synergistically and produce better pain relief. Paracetamol and / or NSAID should be used unless contraindicated
Paracetamol:Paracetamol works as a weak prostaglandin inhibitor. It achieves this by blocking the production of prostaglandins, thereby blocks pain message to the brain and at source of pain, hence has different action from Aspirin and NSAIDs (non-steroidal anti-inflammatory drugs). Paracetamol can be used by patients for whom NSAIDs are contraindicated, including those with asthma or peptic ulcers. Because there are few interactions with other medications, paracetamol can be taken by people with sensitivity to aspirin. Standard safe dose is 1000mg four times a day or upto 4 gm/day in a normal healthy adult.
NSAIDs: Use any NSAID that the patient has found effective and tolerated well in the past, otherwise consider ibuprofen to the maximal dose. Ibuprofen, 400 mg four times a day (daily maximum = 3,200 mg) Note that the potential adverse effects of chemotherapy, such as hematologic, renal, hepatic, and cardiovascular toxicities, can be increased by the concomitant prescription of NSAIDS. Opioid analgesics are a safe and effective alternative analgesic to NSAIDs.
Monitoring for NSAID toxicities:
- Baseline blood pressure, BUN, creatinine, liver function studies [alkaline phosphatase, LDH, SGOT, SGPT], CBC, and fecal occult blood
- Repeat every 3 mths to ensure lack of toxicity
NSAID and toxicities: Age > 60 y, compromised fluid status, multiple myeloma, diabetes, interstitial nephritis, papillary necrosis, and concomitant administration of other nephrotoxic drugs (including cyclosporine, cisplatin) and renally excreted chemotherapy.
Treatment of toxicity :
- If patient develops gastrointestinal upset, peptic ulcer , hemorrhage or vomiting, consider discontinuing NSAID or changing to selective COX-2 inhibitor. COX-2 inhibitors are associated with lower incidence of GI adverse effects and do not inhibit platelet aggregation, however, they have not been demonstrated to have reduced renal adverse effects.Consider adding antacids, H2 receptor antagonists, omeprazole.
- Discontinue NSAID if liver function studies increase 1.5 times the upper limit of normal.
- If congestive heart failure or hypertension develops or worsens, history of cardiovascular disease, discontinue NSAID.
- Risk of bleeding may significantly increase in patients on anticoagulants, such as warfarin or heparin, if NSAIDs are given.
Guidelines for prescribing opioids in Cancer Pain:
1.Intent:
- To lay down guidelines for
- the use of opioid analgesics in patients with cancer pain
- the use of rescue doses for breakthrough pain
- Conversion / rotation from one opioid to another opioid
- Conversion of oral to transdermal route
- the management of opioid induced side effects
2.Scope of these guidelines
- To standardize prescription of opioids across the service & hospital
- To prevent and actively manage opioid related side effects
3.General principles of opioid prescription
- The WHO analgesic ladder should be followed while treating pain in patients with cancer
- Start treatment at the WHO analgesic ladder step appropriate to the severity of pain
- Oral route preferred. Other routes (S.C, I.V, Transdermal, buccal, transmucosal, PR) can be used when indicated, if oral route cannot be used
- Intramuscular route not recommended as it is painful and unreliable
- Analgesia should be prescribed on a "regular" and not on an "as required" basis
- Appropriate analgesia should be prescribed for breakthrough pain 48
- Mixed agonists-antagonists have limited usefulness in cancer pain and should not be prescribed in combination with opioid agonists. Could precipitate withdrawal in opioid dependent patients
- Reassure patients about the low probability of addiction to opioids & encourage to adhere to treatment regime
4.Principles of opioid maintenance therapy
- Extended release or long-acting preparations on a regular schedule to provide background analgesia once dose requirements are stable
- Rescue doses (1/6th of the 24 hr dose of morphine) in the form of an immediate acting/ short acting opioid preparation should be prescribed for breakthrough pain or acute exacerbations related to activity
- The same opioid, if possible, should be used for breakthrough and "around-theclock" dosing
- Increase the dose of the "around-the-clock" preparation if patient requires more than 3 breakthrough analgesic doses
- Dose increase, if necessary Immediate release preparation: after every 24 hours ii. Controlled release or extended release preparations: after 48 hours
- Each dose increment can be set at 33-50% of the pre-existing dose and should be accompanied by a proportionate increase in the rescue dose
- Transmucosal fentanyl should be used only in opioid tolerant patients and initiated with the lowest dose
- For patients who experience inadequate pain relief or unacceptable side effects : Consider Opioid rotation
Conversion from one opioid to another (Opioid rotation):
- For patients with inadequate pain relief and/or intolerable side effects while on strong opioids: Consider switching to a different opioid
- Determine the amount of current opioid taken in last 24 hours that produced good pain relief
- Calculate the equianalgesic dose of the new opioid d. An equianalgesic table should be used when switching to ensure equivalency
- Safe to reduce the dose of the new opioid by 25% to 50% when switching, if pain relief was good
- If previously pain relief was inadequate, may begin with 100% of equianalgesic dose or increase by 25%
- Assess regularly for efficacy & side effects
Principles for prescription of Transdermal fentanyl:
- Pain should be well controlled on a short acting opioid prior to starting a Transdermal fentanyl patch
- Should be used only in opioid tolerant patients 49
- Not recommended for unstable or poorly controlled pain
- Fever and use of warming devices (warming blankets etc) accelerate absorption from the patch, hence are contraindications for its use
- Analgesic duration is usually 72 hours, but some patients require replacement every 48 hours
Conversion from morphine / codeine to transdermal fentanyl:
- Select mcg/hr dose of transdermal fentanyl based on the last 24 hr dose of Morphine or codeine
- An "as- required" dose of immediate release/ short acting morphine should be prescribed and will be needed particularly during the first 8 to 24 hours
- The patch dose can be increased after 3 days based on the amount of daily 'asrequired" opioid needed
Management of side effects:
- Constipation
- Should prescribe a stimulant laxative stool softener (Liquid paraffin + Na picosulphate)
- Increase dose when increasing dose of opioids
- Adequate fluid and dietary fiber intake
- Add another laxative, e.g.; lactulose 30-60 ml/day, if constipation persists
- Severe constipation: Sena compounds (Senasoft) initiate with 1hs.
- Enema, if fecolith
2.Nausea
- Prophylactic use of anti-emetics may be recommended in emesis prone patients
- If nausea develops;
- Rule out other causes (constipation, CNS pathology, Chemotherapy, radiation therapy, hypercalcemia)
- Consider metoclopramide 10 mg PO every 6-8 hrs, or in refractory cases, haloperidol 0.5-1 mg PO every 6-8 h may be added
- If nausea present despite above, add an "around-the-clock" serotonin antagonist e.g.; ondansetron 8 mg PO TDS.
3.Pruritis
- Assess for other causes (other medications)
- Antihistamines to be used. Diphenhydramine (Benadryl ) 25-50 mg IV or PO every 6 hrs or promethazine (phenargan)12.5 -25mg PO every 6 hrs
- If persisting despite above, consider continuous infusion of naloxone 0.25 mcg/kg/h and titrate up to 1 mcg/ kg/ hr without decreasing analgesic efficacy
4.Sedation
- Assess for other causes (CNS pathology, other medications, hypercalcemia, dehydration, sepsis, hypoxia)
- Reduce dose of opioid
- Consider rotation
- Increase non-opioid analgesics where feasible
- CNS stimulants e.g.; Caffeine 100-200 mg PO every 6 hrs could be considered
5.Respiratory depression
- Shallow breathing & unresponsiveness. Consider naloxone infusion. Dilute 1 ampoule (0.4 mg/ml) in 9 ml of normal saline to a total volume of 10 ml. Give 1-2 ml (0.04-0.08 mg) every 30-60 seconds until symptoms improve.
- If no improvement, consider other causes for change in CNS status
6.Delirium
- Assess for other causes (e.g.; hypercalcemia, CNS metastases, other psychoactive medications)
- Reduce dose of opioid if feasible
- Haloperidol 0.5-2 mg PO or IV every 4-6 hrs
7.Cognitive impairment
- Stable doses of opioids (> 2 weeks) unlikely to interfere with psychomotor & cognitive function.
- Monitor during initial titration
8.Usually tolerance develops for all side-effects with time, except constipation. If persists, consider opioid rotation or maximize non-opioid / neuraxial analgesics/ neuroablative procedures
9.Co-morbidities
Liver disease
- All opioids are metabolized in the liver
- Opioid clearance reduced, bioavailability and half-life increased
- Risk of adverse effects because of higher-than-expected plasma concentrations
- Metabolism of morphine not significantly altered
- Avoid use of Acetaminophen in these patients
Renal disease
Active metabolites of propoxyphene ( norpropoxyphene), morphine (morphine-6-glucuronide [M6G], morphine-3-glucuronide [M3G] and nor morphine) and codeine may accumulate 51
- High levels of M3G may cause hyperalgesia and myoclonus
- Use opioids with caution
- Reduced dose and / or
- Reduce frequency of administration
- Avoid using morphine and codeine in patients with renal failure
- Fentanyl, Alfentanil and Buprenorphine are the safest opioids in patients with chronic renal disease stage 4 or 5 (GFR <30 ml/min). Transdermal fentanyl is the opioid of choice
Dysphagia, Intestinal obstruction: Transdermal Fentanyl patches are the treatment of choice. Start with the lowest strength patch (12.5 mcg) in opioid naïve patients
Basic pharmacology of opioids (available in India)
| Drug | Onset of pain relief | Time to peak pain relief | Duration of pain relief | Bioavailability (range) | Peak plasma level | Half life |
| Codeine 3 | 30-60 min | 45-60 min | 4-6 hr | 40%(12-84%) | 1-2 hr | 2.5-3.5 hr |
| Tramado | 30 min | 180 min | 4-6 hr | 75% | 2 hr | 6 hr |
| Morphine | 20-30 min | 60-90 min | 3-6 hr | 35% (15-64%) | 15-60 min | 1.5-4.5 hr |
Oral to parenteral opioid equivalences table
| OPIOID | Dose (mg) equianalgesic to 10 mg Morphine | IV to PO FACTOR | Duration Of Action | |
| PARENTERAL DOSE | ORAL DOSE | |||
| Codeine | 130 mg | 200 mg | 1.5 | 3-4 hr |
| Morphine | 10 mg | 30 mg | 3 | 3-4 hr |
| Tramadol | 50-100 mg | 3-7 hr | ||
Dose conversion table from other opioids to Transdermal fentanyl
| Transdermal Fentanyl | Morphine | Codeine | ||
| IV/Sub cut | Oral | IV/Sub cut | Oral | |
| 25 mcg/h | 20 mg/day | 60 mg/day | 130 mg/day | 200 mg/day |
| 50 mcg/h | 40 mg/ day | 120 mg/day | 260 mg/day | 400 mg/day |
| 75 mcg/h | 60 mg/day | 180 mg/day | 390 mg/ day | 600 mg/ day |
| 100 mcg/h | 80 mg/ day | 240 mg/day | 520 mg/day | 800 mg/day |
Dose conversion ratios (While switching opioids)
| Current opioid (oral) | New opioid (oral) | How to calculate 24 hour dose |
| Codeine | Morphine | Divide by 10 |
| Tramadol | Morphine | Divide by 5 |
Guidelines for prescribing adjuvants:
What is an adjuvant? An adjuvant is a medication that is not primarily designed to control pain, but can be used for this purpose. They are a diverse group of drugs that includes antidepressants, anticonvulsants (antiseizure drugs), and others.
Antidepressants: The best studied adjuvant analgesics are the tricyclic antidepressants (TCAs), such as amitriptyline , and desipramine. These drugs have been shown to relieve pain independent of their effects on depression; that is, patients who are not depressed may experience pain relief. Once the correct dose is found for the individual patient, favorable results are usually seen within a week; however, side effects, including weight gain, dry mouth, blurred vision and constipation, are possible. They are prescribed either alone or with anticonvulsant in patients with neuropathic pain
Anticonvulsants: Drugs that are primarily used to treat epilepsy (seizures) have been used to treat nerve pain conditions. Many anticonvulsant drugs have been shown in clinical studies to be effective. The most common side effects associated with these drugs are mental clouding and sleepiness. Gabapentin or pregabalin are two agents which are used commonly.
Alpha-2-Adrenergic Agonists: Currently, there are two alpha-2-adrenergic agonists that have some evidence as pain relievers: tizanidine and clonidine
Local Anesthetics: Intravenous infusion of a local anesthetic is a special technique that may be used by pain specialists. Mexilitine has shown to be effective in chronic nerve pain syndromes with 53 lancinating type of pain. Other oral local anesthetics are also used. Lignocaine transdermal patch 5% is used in post herpetic analgesia
Steroids: Corticosteroids can be used as an effective analgesic for treating some cancer pain syndromes. Used in pain due to raised intracranial tension, nerve compression, epidural spinal cord compression, pain due intestinal obstruction, plexopathies and complex regional pain syndrome (reflex sympathetic dystrophy).
Other Adjuvants: -
- Baclofen as a muscle relaxant, is also used to treat nerve pain syndromes.
- Drugs that block wide dynamic range neurons involved in the experience of pain, the Nmethyl-D-aspartate (NMDA) receptor, may also be used as very useful analgesic in refractory neuropathic pain conditions. Examples: Ketamine (anesthetic), dextromethorphan (the cough suppressant, but at higher doses than those needed to block cough)
- Bisphosphonates: is considered for use as part of the regime to treat pain in patients with metastatic bone pain
Guidelines for the management of Neuropathic Pain:
Aim:
- To lay down guidelines for the
- Assessment of neuropathic pain in patients with cancer
- Appropriate treatment regimens based on the underlying mechanisms causing pain
Scope of these guidelines:
- Help clinicians to assess and manage neuropathic pain in cancer effectively
- To minimize risk of side effects due to analgesics
- To standardize prescriptions and procedures
- To individualize treatment with the goal of maximizing function & quality of life
What is neuropathic pain?
- Pain arising as a direct consequence of a lesion or a disease affecting the somatosensory system (Neuropathic Pain Working Group 2006)
- Common neuropathic syndromes in cancer
- Brachial plexus neuropathies
- Chemotherapy-induced neuropathy: Cisplatin, Oxaliplatin, Paclitaxel, Thalidomide, Vincristine, Vinblastine
- Cranial neuropathies, Post herpetic neuropathy
- Post-radiation plexopathy
- Surgical neuropathies 54
- Phantom limb
- Post-mastectomy syndrome
- Post-thoracotomy syndrome
How to diagnose neuropathic pain?
In the pain clinic, assessment of a pain patient with suspected neuropathic pain aims at recognition of neuropathic pain, localizing the lesion, and diagnosing the causative disease or event. Nerve compression has been reported to be the most common cause of neuropathic pain in cancer patients (79%), followed by nerve injury (16%) and sympathetically mediated pain (5%).
Description/ Characteristics of pain:
- Spontaneous (Stimulus independent):
- burning
- shooting
- stabbing
- Electric shock-like pain
- Pin & needles
- Dysaesthesias (abnormal and unpleasant sensations)
- Paraesthesias (abnormal, but not unpleasant sensations)
Stimulus evoked pains (Elicited by mechanical, thermal or chemical stimulus):
- Hyperalgesia (increased response to normally painful stimulus)
- Mechanical allodynia (pain from non-painful stimuli)
- Inability to tolerate cloth, air from fan or air conditioning vent touching the skin
- Cold allodynia (pain evoked by cold stimulus)
Pain assessment tools:
Pain Detect Questionnaire: This simple scoring system is available in English, Hindi, Marathi and other many Indian languages, can be completed by individual patients, helped by their relatives. A diagnosis of neuropathic pain can be made. A total of score points are estimated out of 35 points. < 12 : negative , 13-18: possible, > 19 : positive neuropathic pain.
Frequency of assessments: Review regularly, at least once a week, to determine the effectiveness of treatment. If patient is in hospital, more often twice a week.
Management of neuropathic pain:
- Initiate pharmacotherapy with only one drug at a time 55
- Neuropathic pain can be treated by unconventional analgesics (adjuvants) which are primarily not analgesics e.g. antidepressants, anticonvulsants or combined with conventional medications such as opioids.
- Adjuvants analgesics e.g. tricyclic antidepressants (TCAs) and anticonvulsants are used as first line drugs.
- If pain relief is inadequate with one drug, TCA+ anticonvulsants (Gabapentin or pregabalin) may be used.
- Selective serotonin reuptake inhibitors (SSRI) or selective nor epinephrine reuptake inhibitors (SNRI) may be added as 2nd line drugs e.g. duloxitine, lamotrigine.
- Aggressive side effects management is required to open the therapeutic window.
- Opioids may be added as third line drug if the combination fails to provide satisfactory relief.
- Capsaicin cream, lidocaine 5% patch or Inj ketamine may also be used to obtain optimum pain relief. The starting dose and any titration of each pharmacological intervention should be properly planned in individual patient, taking into consideration the potential side-effects and interactions with other medication.
- Non-Opioids: NSAIDs and Paracetamol have a limited use in neuropathic pain.
- However patient may have associated nociceptive pain which may respond to the same.
- Interventional treatments for neuropathic pain should be offered to any patient whose pain is not adequately treated with first and second line drugs or for patients with intolerable side-effects from medication.
- Invasive treatments should usually be carried out as part of a multidisciplinary treatment plan
Guidelines for management of bone metastasis pain:
INTRODUCTION:
- Cancer-induced bone pain (CIBP) is one of the most common types of cancer pain, present in 28-45% of patients with bone metastasis. CIBP often results in hospice or hospital admission and is associated with reduced quality of life, increased psychological distress and decreased physical and social functioning.
- The degree and location of bone metastases do not necessarily correlate with the severity of pain. Some patients have widespread bone metastases but minimal pain, whereas others have minimal bone metastases but severe pain. Apart from CIBP, the other complications of cancer-induced bone disease are hypercalcaemia, cytopaenias and risk of pathological fractures.
- CIBP does not exist as a single entity, but is instead a combination of background pain and breakthrough pain.
- Almost half of breakthrough pain episodes were rapid in onset (<5 min) and short in duration (<15 min). Forty-four per cent of patients with breakthrough pain had pain that was unpredictable e) Up to 45% of patients with CIBP report poor pain control
DIAGNOSIS:
A cancer patient requires a comprehensive evaluation to determine the etiology and site of the specific pain syndrome involved bone metastasis.
- Plain X-ray: Lytic, nodular or rounded, well-circumscribed sclerotic lesions are seen on a plain x-ray.
- CT-scan: Is extremely useful to confirm isolated lesions, assess the extent of soft tissue involvement and to differentiate between metastasis and degenerative changes.
- Bone scan: Is a sensitive imaging tool to detect bone metastases early and still the optimum method for diagnosis. However, small deposits (less than 2 mm) may be missed.
- MRI: Useful to image the spine, vertebral bodies, paraspinal area including the soft tissue component and spinal cord compression. It can differentiate between benign and malignant vertebral collapse4 .
- PET-Scan: Positron emission tomography uses radioactive F-fluorodeoxyglucose (FDG) as a tracer to highlight metabolically active cells. It is not specific for bone metastases and will also identify any area with increased metabolic rate and glucose turnover. The test is now used as an investigational tool for most cancers5 .
- Biochemical markers: The biochemical markers currently available lack specificity and are of no value in the diagnosis of skeletal metastasis. They include: alkaline phosphatase, urinary hydroxyproline and urinary hydroxyproline: creatinine ratio.
TREATMENT:
- The current treatment options for CIBP are wide-ranging and include external beam radiotherapy, opioid analgesia, non-steroidal anti-inflammatory drugs (NSAIDs) and bisphosphonates. In addition, local surgery and anaesthetic techniques are used. However, each of these treatment options is accompanied by limitations in their use.
- Radiotherapy is the gold standard treatment of CIBP. Complete pain relief is only achieved in about 25% of patients, whereas 50% of patients will achieve 50% pain relief .
- NSAIDs, being anti-inflammatory drugs are important in the treatment of CIBP based on experience rather than strong evidence.
- Opioids are an effective therapy for background pain in CIBP. However, a normal release morphine will probably be ineffective in patients with rapid-onset, short duration breakthrough pain due to its slow onset of action.
- Alternative routes of fast acting opioids are transnasal(butorphenol),transmucosal (fentanyl).
- Bisphosphonates are used to reduce skeletal morbidity from bone metastases and for analgesia in CIBP.
- Pain produced by bone metastases influences the nervous system peripherally and centrally. Spontaneous breakthrough pain which may occur at rest in bone metastasis, may poorly respond to opioids.
Guidelines for the management of Breakthrough Pain (BTP) in cancer:
Aim:
To lay down guidelines for
- Identification & assessment of breakthrough pain in cancer patients.
- Appropriate treatment regimens for the management of such pain
- To reduce the intensity, severity & impact of each bout of pain
Definition: A transitory exacerbation of pain experienced by the patient who has a relatively stable and adequately controlled baseline pain (Portenoy et al 2004). Prevalence of BTP is 20- 65%.
What are the types of BTP?
- Incident pain: precipitated by movement/coughing
- Spontaneous pain : occurs in absence of a specific trigger & at random Should be differentiated from end-of-dose pain which occurs:-
- just prior to the scheduled dose of analgesia
- Either due to an inadequate analgesic dose or too long an interval.
- Gradual onset.
How to identify breakthrough pain?
- Rapid onset
- Severe intensity
- Variable duration (usually 30 minutes)
Assess
- All patients with cancer pain for presence of BTP (D)
- Each episode of breakthrough pain for;
- Frequency
- Duration
- Pain intensity
- Precipitating factors
- Baseline pain treatments and their effectiveness
- Re-assess to determine;
i Efficacy & tolerability of treatment.
ii. Change in nature of the pain
Management of BTP
- Optimize the round-the-clock analgesia by titrating opioids
- Should always prescribe rescue analgesia and teach the patient how to use it.
- Opioids are the rescue medications of choice. Little evidence to support the use of non-opioid analgesics for breakthrough pain.
- The opioid should be chosen based on its pharmacokinetic properties. If possible, rescue dose should be of the same opioid used for baseline pain (Mercadante et al 2002) but there are no compelling reasons for using the same opioid.
- European Association of Palliative Care (EAPC) recommendations are to start with 1/6th (17%) of the daily dose, and subsequent titration according to clinical effect.(Hanks et al 2001).
- The dose of opioid rescue medication should be determined by individual titration(Zeppetella, 2006)
- Incident pain: pre-emptive use of a short acting opioid, 30 minutes before the activity which precipitates pain. (Mc Carberg, 2007).
- Spontaneous pain: Oral transmucosal fentanyl citrate (OTFC) found to be an effective treatment for breakthrough pain by a recent Cochrane review. (Zeppetella, 2006)
- End-of-dose failure: alter the around-the-clock medication to increase the dose or shorten the dosing interval (McCarberg, 2007).
- Transdermal fentanyl: duration of action- 72 hours. In some patients (3-43%), duration is between 48-72 hrs & may require breakthrough medications after 48 hrs. Replace the patch in these patients every 48 hrs rather than 72 hrs instead of increasing the dose of the patch.
- More than 4 episodes of breakthrough pain in a day: review the baseline pain management. (Coluzzi, 1998)
Management of BTP:
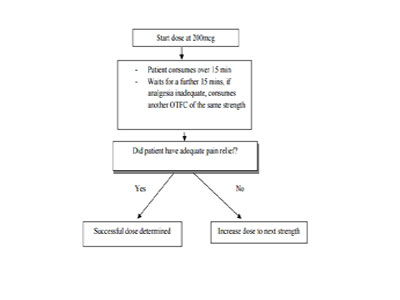
Titration to obtain "correct dose" of oral morphine for breakthrough pain
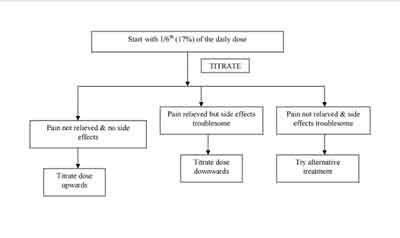
Breakthrough dose of oral morphine for background dose of transdermal fentanyl
| Background dose of transdermal fentanyl (µg/hr) | Breakthrough dose of oral morphine (mg) |
| 25 | < 20 |
| 50 | 25-35 |
| 75 | 40-50 |
| 100 | 55-65 |
| 125 | 70-80 |
| 150 | 85-95 |
| 175 | 100-110 |
| 200 | 115-125 |
| 225 | 130-140 |
| 250 | 145-155 |
| 275 | 160-170 |
| 300 | 175-185 |
Guidelines for interventional Pain management:
Indications:
- Usually nerve blocks are indicated when even oral strong analgesics do not provide even 50% pain relief and there are intolerable side effects.
- can be relieved. Verify interventional technique which will provide sufficient benefit for the diagnosed pain syndrome.
- Pain likely to be relieved with nerve block (eg, pancreas/ upper abdomen with celiac plexus block, lower abdomen with superior hypogastric plexus block, intercostal nerve, or peripheral nerve)
Commonly used interventional procedures:
1) Neurodestructive procedures for well-localized pain syndromes
- Head and neck: peripheral nerve block
- Upper extremity: brachial plexus neurolysis
- Thoracic wall: epidural neurolysis, intercostal neurolysis
- Upper abdominal pain (visceral): celiac plexus block, thoracic splanchnicectomy
- Midline pelvic pain: superior hypogastric plexus block
- Rectal pain: intrathecal neurolysis, midline myelotomy or superior hypogastric plexus block, Ganglion Impar
- Unilateral pain syndromes: cordotomy h) Consider intrathecal L/S phenol block
2) Radiological interventions: Percutaneous vertebroplasty/kyphoplasty, Radiofrequency ablation for bone lesions
3) Neurostimulation procedures for cancer-related symptoms (ie, peripheral neuropathy)
4) Regional infusions (requires spinal intrathecal infusion pump) easy to internalize implanted pump; for infusions of opioids, local anesthetics, clonidine, and ziconotide
Contraindications ;
Infection, coagulopathy, very short or lengthy life expectancy, distorted anatomy, patient unwillingness, medications that increase risk for bleeding,(eg, use of anti-angiogenesis agents such as bevacizumab) or technical expertise for performing interventions is not available.
Guidelines for pain management at end of life:
Introduction:
- Many patients at the end of life are unable to verbally report pain using standardized scales. In this situation, evaluate behavioral cues such as facial grimacing, guarding, or vocalizing.
- Rule out other potential causes of distress such as constipation, urinary distension, or emotional and spiritual distress.
- Administration of opioids may be complicated by the inability to swallow. Alternate routes of administration include buccal, sublingual, rectal, subcutaneous, or intravenous delivery.
- Doses may be decreased in some cases, as organ system failure leads to reduced excretion of the drug and or its metabolites, as well as other factors.
- Myoclonus may occur when higher doses of opioids are administered, particularly in the face of renal dysfunction. Reducing the opioid dose or rotating to another opioid can be effective, and adding benzodiazepines can be helpful
Guidelines for pain management in cancer survivors:
INTRODUCTION:
A large number of disease-free cancer survivors live with pain or neuropathies induced by treatment or by the cancer itself. Sometimes these conditions resolve over time, but irreversible damage to tissue and nerves can cause pain and neuropathy to progress and persist indefinitely. Because health care professionals may not recognize these as delayed problems or know how to identify those at greatest risk, many of these conditions go undiagnosed and untreated. Most chronic pain syndromes and neuropathies experienced by disease-free survivors of cancer originate from;
- an injury to peripheral nerves from surgical trauma,
- neurotoxicity of chemotherapeutic agents
- radiation-induced damage to nerves
Other sources of persistent or intermittent pain include:
- myofascial pain dysfunction syndrome, which is characterized by trigger points in muscle or at the junction of muscle and fascia that refer pain to other areas of the body, and which is associated with breast, thorax, and head and neck surgery in cancer survivors.
- fistula formation following pelvic surgery.
- chronic inflammation, such as radiation-induced enteritis or proctitis.
- osteoradionecrosis,a radiation-induced demineralization and vascularization of the bone.
- Persistent nerve damage from tumor infiltration of nerves.
TREATMENT:
Anticonvulsants, tricyclic antidepressants, and opioids can be effective for alleviating neuropathic pain. Novel pharmacologic agents such as neuroprotective compounds e.g amifostine,glutamine,and glutathione,and neurotrophic factors (such as nerve growth factor) can also be effective. Vitamin E has shown some promise in prophylaxis of 66 chemotherapy- induced peripheral neuropathy with cisplatin and paclitaxel. Less is known about nonpharmacologic and alternative therapies.
To read full guidelines click on the following link:
https://clinicalestablishments.nic.in/WriteReadData/329.pdf

Disclaimer: This site is primarily intended for healthcare professionals. Any content/information on this website does not replace the advice of medical and/or health professionals and should not be construed as medical/diagnostic advice/endorsement or prescription. Use of this site is subject to our terms of use, privacy policy, advertisement policy. © 2020 Minerva Medical Treatment Pvt Ltd