- Home
- Editorial
- News
- Practice Guidelines
- Anesthesiology Guidelines
- Cancer Guidelines
- Cardiac Sciences Guidelines
- Critical Care Guidelines
- Dentistry Guidelines
- Dermatology Guidelines
- Diabetes and Endo Guidelines
- Diagnostics Guidelines
- ENT Guidelines
- Featured Practice Guidelines
- Gastroenterology Guidelines
- Geriatrics Guidelines
- Medicine Guidelines
- Nephrology Guidelines
- Neurosciences Guidelines
- Obs and Gynae Guidelines
- Ophthalmology Guidelines
- Orthopaedics Guidelines
- Paediatrics Guidelines
- Psychiatry Guidelines
- Pulmonology Guidelines
- Radiology Guidelines
- Surgery Guidelines
- Urology Guidelines
Case of Diamond–Blackfan anemia: a report

Dr Colin Byron Noel at Department of General / Transplant Surgery, University of Cape Town and Groote Schuur Hospital, Cape Town, South Africa has reported a case of Diamond–Blackfan anemia that has been published in Journal of Medical Case Reports.
Diamond–Blackfan anemia is a rare congenital red blood cell aplasia characterized by failed erythropoiesis, congenital abnormalities in up to 50% of patients, growth retardation in up to 30% of patients, and a predisposition to malignancy. Diamond–Blackfan anemia is both clinically and genetically a heterogenous condition ranging from subtle asymptomatic erythroid abnormalities to non-immune hydrops fetalis. Current treatment options include corticosteroid therapy, chronic red blood cell transfusions, and hematopoietic stem cell transplantation with gene therapy receiving recent attention.
A 6-week-old Caucasian girl was admitted to a private hospital in South Africa with acute onset symptomatic cardiac failure secondary to anemia. Her parents reported a 1-day history of lethargy, poor feeding, shortness of breath, and irritability on a background history of progressive pallor.
There was no family history of note. Antenatal history included a low maternal pregnancy-associated plasma protein A (PAPP-A) level (0.376 IU/L) which resulted in a high-risk screening protocol for intrauterine growth restriction (IUGR) and fetal chromosomal anomalies. Cell-free fetal deoxyribonucleic acid testing from maternal blood excluded aneuploidies for the common trisomies [10, 11, 12] and subsequent fetal anomaly ultrasound and echocardiogram scans were all normal. A caesarean section was performed at 37 weeks for spontaneous labor, IUGR, and breech presentation. The delivery was uneventful and apart from a low birth weight of 2465 g, a healthy baby was discharged 3 days post caesarean section as per normal protocol.
On admission to hospital at 6 weeks of age, the baby under examination was severely anemic, tachycardic, and lethargic. There were no stigmata of immunocompromise, infection, or icterus. The baby weighed 3200 g with a head circumference of 38 cm.
There were no obvious craniofacial or skeletal abnormalities of note and examinations of her other systems were normal. The preliminary results with normal range for age in brackets showed a hemoglobin (Hb) level of 3.1 gm/dL (10–18 gm/dL) and a hematocrit of 9% (31–55%), mean corpuscular volume of 106 fl (85–123 fl), mean corpuscular Hb concentration 34 g/dL (32–37 g/dL), reticulocyte production index of 0.0, and an absolute reticulocyte count of 5.1 × 109/L (20–60 × 109/L). Her white cell count was low 4.0 × 109/L (5–19.5 × 109/L) but apart from a low neutrophil count of 0.32 × 109/L (1–9 × 109/L), the remaining differential count was normal. Her platelet count was increased 655 × 109/L (140–420 × 109/L). Her C-reactive protein was marginally raised at 7.7 mg/L (< 5 mg/L), and the infective work up was positive for Escherichia colicultured from the urine. Tests for cytomegalovirus, human immunodeficiency virus (HIV), rubella, Epstein–Barr virus, toxoplasmosis, herpes simplex virus 1 and 2, and parvovirus B19 were all negative. A diagnosis of E. coli urosepsis was made. The baby was transfused with leukodepleted irradiated red cell concentrate to an Hb level of 10 g/dL and given goal-directed antibiotics and discharged 6 days later.
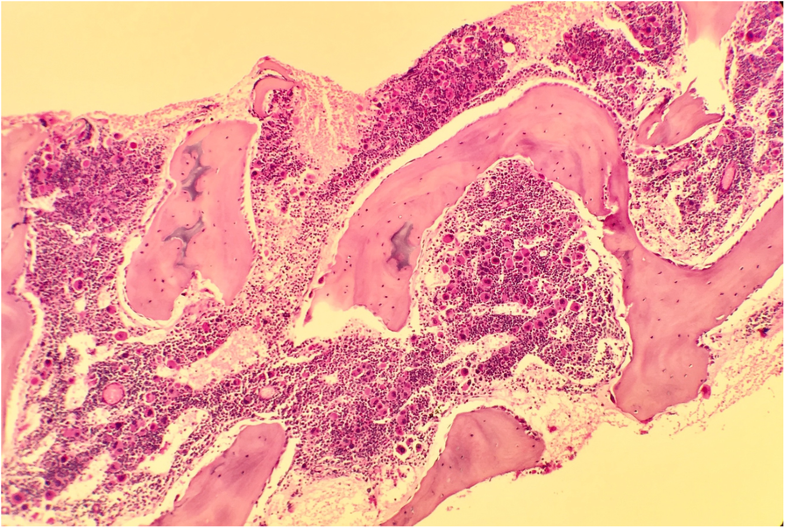 Courtesy Journal of Medical Case Reports
Courtesy Journal of Medical Case ReportsDue to the unavailability of molecular and eADA testing in South Africa, specimens for molecular testing were sent to Oxford, UK, for identification of a possible heterozygous pathogenic variant in one of the genes associated with DBA. A multigene panel using the Multiplex Ligation-dependent Probe Amplification kit (MRC-Holland) confirmed a heterozygous whole gene deletion of RPL35A. In addition, dried plasma was sent to Duke University Medical Center, USA, to exclude adenosine deaminase 2 deficiency.
Once the diagnosis of DBA was made, the non-standardized management and limited experience in managing DBA in South Africa resulted in inconsistencies in opinion in the optimal early management of the case. Controversies around Hb transfusion threshold, optimal Hb target, frequency of transfusions, and timing and dosage of corticosteroid treatment and hematopoietic stem cell transplantation (HSCT) resulted in different opinions from different specialist practitioners.
Together with input from international DBA specialists managing large numbers of patients with DBA, the initial hematopoietic management in this case was directed toward correcting the anemia with transfusions every 3–5 weeks with irradiated leukodepleted RBC concentrate. A transfusion threshold of 8 g/dL was used and a volume of 10–15 ml/kg transfused on each visit. Planned iron chelation therapy to prevent transfusional hemosiderosis will be delayed until approximately 170–200 ml/kg of transfused red packed cells has been given. A planned trial of corticosteroids will be given at 1 year of age.
At follow-up at 6 months of age, the baby was stable requiring red cell concentrate infusion therapy every 3–4 weeks. Persistence in the neutropenia was noted, with no changes in the other cell lineages. Her current ferritin level is 573 μg/L and a total of 90 ml/kg of red packed cells has been transfused, thus, iron chelation therapy has not yet been instituted. Apart from a delay in gross motor development and growth (weight and height), all other parameters and development are within normal limits.

Disclaimer: This site is primarily intended for healthcare professionals. Any content/information on this website does not replace the advice of medical and/or health professionals and should not be construed as medical/diagnostic advice/endorsement or prescription. Use of this site is subject to our terms of use, privacy policy, advertisement policy. © 2020 Minerva Medical Treatment Pvt Ltd