- Home
- Editorial
- News
- Practice Guidelines
- Anesthesiology Guidelines
- Cancer Guidelines
- Cardiac Sciences Guidelines
- Critical Care Guidelines
- Dentistry Guidelines
- Dermatology Guidelines
- Diabetes and Endo Guidelines
- Diagnostics Guidelines
- ENT Guidelines
- Featured Practice Guidelines
- Gastroenterology Guidelines
- Geriatrics Guidelines
- Medicine Guidelines
- Nephrology Guidelines
- Neurosciences Guidelines
- Obs and Gynae Guidelines
- Ophthalmology Guidelines
- Orthopaedics Guidelines
- Paediatrics Guidelines
- Psychiatry Guidelines
- Pulmonology Guidelines
- Radiology Guidelines
- Surgery Guidelines
- Urology Guidelines
Benign Hyperplasia Of Prostate (BPH)-Standard Treatment Guidelines
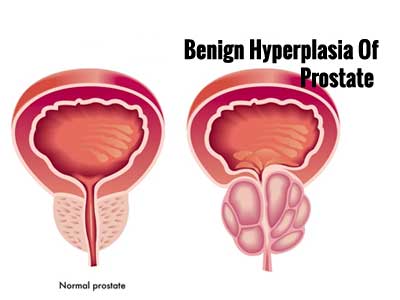
Benign prostatic hyperplasia(BPH) is an enlarged prostate gland. The prostate gland surrounds the urethra, the tube that carries urine from the bladder out of the body. As the prostate gets bigger, it may squeeze or partly block the urethra. This often causes problems with urinating.
BPH is a common problem that affects the quality of life in approximately one third of men older than 50 years. BPH is histologically evident in up to 90% of men by age 85 years.
Worldwide, approximately 30 million men have symptoms related to BPH. It tends to be more severe and progressive in African-American men because of the higher testosterone levels, 5-alpha-reductase activity, androgen receptor expression, and growth factor activity in this population
Ministry of Health and Family Welfare, Government of India has issued the Standard Treatment Guidelines for Benign Hyperplasia Of Prostate (BPH).
Following are the major recommendations :
Use of the term benign hyperplasia of the prostate (BPH) - BPH is a histological diagnosis. It symptoms come under the umbrella of lower urinary tract symptoms. While BPH occurs in the vast majority of elderly men, it can be difficult or impossible to directly attribute the symptoms of the patient to BPH.
Introduction
Line 17-20 : ”However, a significant proportion, particularly in rural areas tend to disregard symptoms till complications develop and this segment requires a more proactive and individualized approach.” There is no evidence that the rural population disregard symptoms more than the urban people. The lack of access to effective health care in rural areas will make a proactive and individualized approach difficult. It is important for guidelines to be applicable to the entire populations of the country.
Evaluation of BPH
Symptom assessment – Need to state the complete term International Prostatic Symptom Score before using the abbreviation IPSS.
Mandatory diagnostic tests
- PSA measurement – Need to state the complete term. There is no evidence that prostate specific antigen measurement has any role in the routine management of BPH. The role of PSA screening for prostate cancer is controversial. Metaanalysis of the recent evidence showed screening to have no significant impact on either overall mortality or death from prostate cancer with significant overdiagnosis and overtreatment and is unlikely to save lives. It cannot be recommended in India. A patient anxious about prostate cancer should be explained about the benefits and risks of PSA screening and about the available evidence. He should be then in a position to make an informed choice.
- Abdominal ultrasound evaluation –Upper tract imaging is not required for routine evaluation of BPH. The estimation of prostate size by abdominal ultrasound is also unreliable. Ultrasound evaluation should be restricted to the bladder and post void residual urine as an optional test.
- Uroflowmetry -This is not mandatory even in developed countries. The equipment is specialized, expensive (Indian makes cost Rs. 50000/- approx., foreign equipment -1.5 -2.5 lacs/- approx.) and require regular maintenance and calibration. It cannot be mandatory and should be an optional test.
Optional diagnostic tests
Blood urea – Blood urea estimation is superfluous when creatinine is being measured. It is not as accurate indicator of renal function as creatinine. Unnecessary.
Urine Cytology – Urinary cytology is a specialized test with low sensitivity. In absence of microscopic haematuria, it is unlikely to be useful.
Urethrography – An uroflowmerty is a non invasive test which can indicate a possible urethral stricture. An urethrography is invasive and will require prior urine culture sensitivity before its performance.
Uroflowmetry and ultrasound abdomen can be added as optional tests.
Treatment options
“Dose titration is not essential for Alfuzosin and Tamsulosin ” should read - Dose titration is not required for extended release Alfuzosin and Tamsulosin. “Hypotensive episodes are least seen with use of Alfuzosin” – The vasodilatory effects of Alfuzosina nd Tamsulosin are similar. There are no statistical differences between the two in this regard [4].
Alpha reductase inhibitors: “Reduction of blood loss during TURP is evident
only after long term use of 5 ARI’s.” - The evidence is very weak and not enough
for a recommendation[5,6,7,8].
Combination therapy: “Concominant use of Alpha adrenergic blockers and 5Alpha reductase inhibitors is appropriate therapy for patients with LUTS due to BPH, particularly if response has been insufficient with either drug.” - Combination therapy is necessary in those who are at risk of progression (moderate to severe LUTS, enlarged prostates, and reduced Qmax) . Response is not a criterion for combination therapy [9].
Indications for surgery
“Patients presenting with chronic low pressure require catheterization and urodynamic evaluation” – Catheterization is not required unless the patient has acute on chronic retention, overflow incontinence or obstructive uropathy with raised creatinine.
Minimally invasive procedures
TUNA and intraprostatic stents – Cannot be recommended based on current evidence.
Surgical (endoscopic and open) procedures
- “Open prostatectomy is still an acceptable procedure for glands exceeding 100 gms in wt.” – Any gland over 60 gms can be managed with open prostatectomy if TURP is not available.
- “Follow-up with IPSS, DRE & PSA recommended every 3 to 6 months initially and annually thereafter.” – Three monthly follow up is not required after surgery. PSA is certainly not needed.
Conclusions: Very poorly written clinical practice guideline. It is unfit for use in its present form. Needs extensive revision.
Guidelines by The Ministry of Health and Family Welfare :
Prof. Nitin S. Kekre, Professor and Head
Dr. Arabind Panda, Associate Professor
Department of Urology
CMC Medical College, Vellore
NOTE from the author- This standard treatment guideline on BPH does not state the methodology used to arrive at the recommendations. The levels of evidence are not mentioned. There is no grading of the recommendations. The document is without any references to back up the claims. The authors should be clearly mentioned in the published version ( we understand this is not desirable in a document for peer review). A conflict of interest declarations should be included in the final document.

Disclaimer: This site is primarily intended for healthcare professionals. Any content/information on this website does not replace the advice of medical and/or health professionals and should not be construed as medical/diagnostic advice/endorsement or prescription. Use of this site is subject to our terms of use, privacy policy, advertisement policy. © 2020 Minerva Medical Treatment Pvt Ltd