- Home
- Editorial
- News
- Practice Guidelines
- Anesthesiology Guidelines
- Cancer Guidelines
- Cardiac Sciences Guidelines
- Critical Care Guidelines
- Dentistry Guidelines
- Dermatology Guidelines
- Diabetes and Endo Guidelines
- Diagnostics Guidelines
- ENT Guidelines
- Featured Practice Guidelines
- Gastroenterology Guidelines
- Geriatrics Guidelines
- Medicine Guidelines
- Nephrology Guidelines
- Neurosciences Guidelines
- Obs and Gynae Guidelines
- Ophthalmology Guidelines
- Orthopaedics Guidelines
- Paediatrics Guidelines
- Psychiatry Guidelines
- Pulmonology Guidelines
- Radiology Guidelines
- Surgery Guidelines
- Urology Guidelines
Antiphospholipid syndrome in man with severe pulmonary hypertension
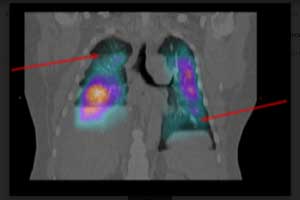
A case report of Antiphospholipid syndrome in a man with severe pulmonary hypertension has been published in the journal Annals of the American Thoracic Society. Most cases of APS (80%) are in women.
Antiphospholipid syndrome (APS) is an autoimmune disorder which occurs when the immune system mistakenly produces antibodies that make host blood much more likely to clot. It can be caused by an underlying condition, such as an autoimmune disorder, infection or certain medications.
APS is classified as primary or secondary, depending on its association with other autoimmune disorders. Primary APS is diagnosed in patients demonstrating the clinical and laboratory criteria for the disease without other recognized autoimmune disease. Secondary APS is diagnosed in patients with other autoimmune disorders, such as systemic lupus erythematosus (SLE).
According to history a 43-year-old man with a history of hypertension and obesity presented to his primary physician with the sudden onset of dyspnea. A contrast-enhanced computed tomographic (CT) scan of the chest revealed bilateral pulmonary emboli (PE), and he was treated with rivaroxaban as an outpatient. His dyspnea continued to progress over the next 6 months despite treatment with anticoagulation.
An initial ventilation-perfusion (V/ Q) scan was obtained because of concern for chronic thromboembolic pulmonary hypertension (CTEPH), given his history of PE, which showed a triple-matched defect in the right lower lung and on single-photon emission CT (SPECT) imaging noted presence of bilateral mismatched perfusion defects.
A closer review of his most recent chest CT imaging revealed interlobular septal thickening in bilateral lung fields, more pronounced on the right, as well as a right-sided pleural effusion. In addition, the CT demonstrated abnormal cardiac morphology with interventricular septal flattening as well as dilatation of the right atrium (RA) and right ventricle (RV). An
His ECG confirmed severe RV dysfunction, with a severely dilated and hypokinetic RV and normal left ventricular function. An emergent right heart catheterization revealed a right atrial pressure of 23 mm Hg, mean pulmonary artery pressure of 54 mm Hg, with a pulmonary artery wedge pressure of 14 mm Hg and a cardiac index of 1.6 L/ min/m2.
Laboratory values obtained at admission included brain natriuretic peptide and D-dimer levels, which were elevated at 355 pg/ml and 4.78 mg/ml, respectively. The international normalized ratio was elevated to 1.97, and prothrombin time was elevated to 23.4 seconds while on rivaroxaban therapy. Differential diagnosis at this time included CTEPH, idiopathic pulmonary arterial hypertension (PAH), and other causes of PAH related to connective tissue disease or pulmonary venoocclusive disease (PVOD).
The patient was transferred to ICU, and intravenous dobutamine was started for inotropic support. Subsequently, intravenous epoprostenol was added for presumed severe PAH and impending RV failure. Within 2 hours of initiating epoprostenol infusion at 2 ng/kg/min, the patient developed significant hypoxemia and acute pulmonary edema, prompting increased concern for PVOD. Epoprostenol infusion was immediately discontinued and the patient was initiated on diuretic therapy, with improvement in pulmonary edema.
Serological workup revealed a positive antinuclear antibody test (1:320, homogenous pattern), positive lupus anticoagulant, abnormal dilute Russell viper venom test, and elevated levels of anticardiolipin immunoglobulin G and b2 glycoprotein antibodies.
On the basis of these findings, the patient was diagnosed with antiphospholipid syndrome (APS). He remained on intravenous dobutamine and was started on warfarin and hydroxychloroquine. His clinical course stabilized on this regimen while in the ICU.
For more details click on the link: https://doi.org/10.1513/AnnalsATS.201802-092CC

Disclaimer: This site is primarily intended for healthcare professionals. Any content/information on this website does not replace the advice of medical and/or health professionals and should not be construed as medical/diagnostic advice/endorsement or prescription. Use of this site is subject to our terms of use, privacy policy, advertisement policy. © 2020 Minerva Medical Treatment Pvt Ltd