- Home
- Editorial
- News
- Practice Guidelines
- Anesthesiology Guidelines
- Cancer Guidelines
- Cardiac Sciences Guidelines
- Critical Care Guidelines
- Dentistry Guidelines
- Dermatology Guidelines
- Diabetes and Endo Guidelines
- Diagnostics Guidelines
- ENT Guidelines
- Featured Practice Guidelines
- Gastroenterology Guidelines
- Geriatrics Guidelines
- Medicine Guidelines
- Nephrology Guidelines
- Neurosciences Guidelines
- Obs and Gynae Guidelines
- Ophthalmology Guidelines
- Orthopaedics Guidelines
- Paediatrics Guidelines
- Psychiatry Guidelines
- Pulmonology Guidelines
- Radiology Guidelines
- Surgery Guidelines
- Urology Guidelines
Antimicrobial Therapy in Patients with Severe Sepsis and Septic Shock in ICU: Guidelines

ICMR has developed the Principles of Initial Empirical Antimicrobial Therapy in Patients with Severe Sepsis and Septic Shock in The Intensive Care Units. Following are its major recommendations
Definitions
Systemic inflammatory response syndrome (SIRS)
Two or more of the following variables
- Fever > 38°C (100.4°F) or hypothermia < 36°C (96.8°F)
- Tachypnea (>20 breaths/min) or PaCO2 < 32 mmHg
- Tachycardia (heart rate >90 beats/min)
- Leukocytosis or leucopenia : WBC > 12,000 cells/mm3, <4,000 cells/mm3 or > 10% immature band forms
Sepsis : Systemic inflammatory response syndrome that occurs due to a “known or suspected” pathogen (bacteria, viruses, fungi or parasites)
Indian Council of Medical Research, Department of Health Research has issued the ICMR Antimicrobial guidelines for Principles of Initial Empirical Antimicrobial Therapy in Patients with Severe Sepsis and Septic Shock in The Intensive Care Units. Following are the major recommendations :
Severe sepsis
Sepsis plus evidence of organ dysfunction or tissue hypoperfusion as follows –
- Altered mental status.
- Hypoxemia, with PaO2/FIO2 <250
- Thrombocytopenia < 100,000/cmm
- Bilirubin >2mg/dl
- INR >1.5 or aPTT> 60 seconds.
- Urinary output of 0.5 ml/kg for at least 2 hours or Serum creatinine >2mg/dl despite fluid resuscitation.
- Tissue hypoperfusion as suspected by mottled skin, capillary refilling time ≥ 2 seconds or lactate >4 mmol/l
- Hypotension : Systolic blood pressure (SBP) ≤90 mmHg or mean arterial pressure ≤70 mm Hg.
Sepsis induced hypotension SBP <90 mm Hg or MAP <70 mm HG or SBP decrease >40 mm Hg
Septic shock Sepsis induced hypotension that persists despite adequate fluid resuscitation, requiring vasopressors to maintain the blood pressure.
Recently, the definitions have been updated as follows:
Sepsis should be defined as life-threatening organ dysfunction caused by a dysregulated host response to infection. For clinical operationalization, organ dysfunction can be represented by an increase in the Sequential [Sepsis-related] Organ Failure Assessment (SOFA) score of 2 points or more.
In patients admitted from the community or emergency department, it can be assumed that patients had no pre-existing organ dysfunction, baseline SOFA score assumed to be zero. Organ dysfunction can be identified in these patients by the quick SOFA or qSOFA. The presence of any two of respiratory rate ≥22, altered mentation or systolic blood pressure ≤100 mm Hg identified high risk of patients. qSOFA is an extremely useful screening tool for organ dysfunction, especially in patients outside the ICU. It can be used to suspect sepsis and initiate further investigations and treatment.
Septic shock should be defined as a subset of sepsis in which particularly profound circulatory, cellular, and metabolic abnormalities are associated with a greater risk of mortality than with sepsis alone. Patients with septic shock can be clinically identified by a vasopressor requirement to maintain a mean arterial pressure of 65 mm Hg or greater and serum lactate level greater than 2 mmol/L (>18 mg/dL) in the absence of hypovolemia.
Investigations :
Clinical history and Investigations should be directed at diagnosis, assessing the focus of sepsis, the severity of the sepsis and the risk of resistant organisms.
Tests to diagnose Infection
- Hemoglobin
- White blood cell count (total and differential)
- Relevant cultures with gram stain
- Urine (Routine and microscopic examination)
- Chest X-Ray
- Other relevant radiological investigations (ultrasound, CT scan)
- Lumbar puncture when clinically indicated
- Blood and other relevant cultures, sensitivity, MIC testing
- Procalcitonin (PCT), C-reactive protein (CRP)
- Investigations for Tropical Infections (see below)
Tests to diagnose and quantify severity of organ dysfunction
- Renal function tests (SE, BUN, Cr)
- Liver function test (Bilirubin, AST, ALT, ALKP, GGT, PT, INR, PTT)
- Arterial Blood gas analysis
- Serum lactate
Other tests
- ECG, Echocardiography
- Therapeutic drug monitoring
- Blood Glucose.
Patients at risk for infections from resistant organisms include:
Antimicrobial therapy in preceding 90 days
- Current hospitalization of 5 days or more
- High frequency of community or hospital antibiotic resistance
- Immunosuppressive disease or therapy
- Presence of multiple risk factors for Health Care Associated Infections
- Hospitalization for ≥2 days in preceding 90 days
- Residence in nursing home or long term care facility
- Home infusion therapy
- Chronic dialysis within 90 days
- Family member with MDR pathogen
Common Pathogens
Common resistant organisms include:
Gram negative:
Pseudomonas aeruginosa
E. coli
Klebsiella pneumoniae
Acinetobacter spp
Gram Positive:
Methicillin resistant Staphylococcus aureus (MRSA)
Entercoccus faecium
Vancomycin resistant enterocccci
Fungi:
Candida spp
Resistance Patterns: ICMR AMR Data 2014
Table 1. Staphylococcus aureus and Enterococcus ICMR AMR National Data 2014
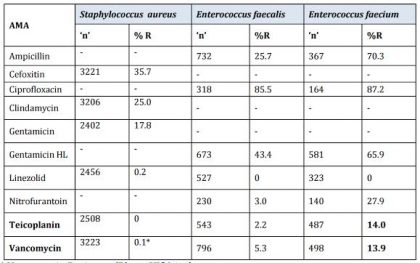
* Vancomycin Resistant (R) are VISA isolates.
Cefoxitin : Surrogate marker for Methicillin.
Table2. Enterobacteriaceae isolates. ICMR AMR National data 2014.
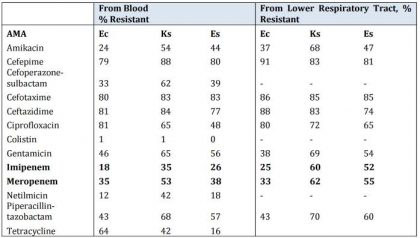
Note: Ec:Escherichia coli; Ks:Klebsiellaspp.; Es : Enterobacter spp.
Table 3. Pseudomonas aeruginosa and Acinetobacter baumannii ICMR AMR National Data 2014
| AMA | Pseudomonas aeruginosa ‘n’ 374 R % | Acinetobacter baumannii ‘n’ 599 R % |
| Amikacin | 35 | 75 |
| Aztreonam | 48 | 87 |
| Cefepime | 41 | 81 |
| Cefoparazone-sulbactam | 38 | 57 |
| Ceftazidime | 47 | 84 |
| Colistin | 10 | 22 |
| Imipenem | 37 | 63 |
| Levofloxacin | 36 | 73 |
| Meropenem | 47 | 62 |
| Netilmicin | 45 | 69 |
| Piperacillin-tazobactam | 46 | 83 |
| Tobramycin | 33 | 58 |
Table 4. Candida spp. isolated at PGIMER, Chandigarh ICMR AMR Data 2014.
| AMA | C. tropicalis ‘n’ 101 % R | C. krusei ‘n’ 98 % R | C . albicans ‘n’ 50 % R | C. pelliculosa ‘n’ 35 % R |
| Amphotericin B | 1 | 5 | 0 | 0 |
| Fluconazole | 4 | 0 | 8 | 0 |
| Voriconazole | 1 | 3 | 8 | 0 |
| Itraconazole | 8 | 4 | 2 | 0 |
| Posaconazole | 1 | 0 | 0 | 0 |
| Caspofungin | 2 | 8 | 0 | 0 |
| Anidulafungin | 1 | 7 | 2 | 0 |
| Micafungin | 0 | 6 | 2 | 6 |
Indian Society of Critical Care Medicine Multi-center Observational Study to evaluate Epidemiology and Resistance patterns of common ICU-Infections (MOSER Study) 2012
Table 5. Ventilator Associated Pneumonia organisms – Resistance Pattern of top 5 organisms
| Organisms | ‘n’ | Pan sensitive | Carbapenem resistant | ESBL positive | Multi-drug resistant | Data NA | Amp C producer | Methicillin Resistant | Vanco resistant |
| Acinetobacter | 88 | 3 | 1 | 2 | 77 | 5 | |||
| Klebsiella | 59 | 1 | 1 | 8 | 47 | 2 | |||
| Pseudomonas | 48 | 8 | 0 | 2 | 30 | 8 | |||
| Staphylococcus | 15 | 5 | 2 | 7 | 1 | ||||
| E. coli | 12 | 1 | 0 | 2 | 8 | 1 |
Table 6. Catheter Related Bloodstream Infection Organisms – Resistance pattern of top 5 organisms
| Organisms | ‘n’ | Pan sensitive | Carbapenem resistant | ESBL positive | Multidrug resistant | Data NA |
| Klebsiella | 18 | 5 | 0 | 4 | 9 | |
| Pseudomonas | 11 | 2 | 1 | 4 | 3 | 1 |
| Acinetobacter | 9 | 3 | 0 | 1 | 7 | |
| E. coli | 9 | 5 | 4 | |||
| Candida | 8 |
4. Initial Empirical Antibiotic Therapy
| Clinical condition | Common pathogens | Empirical AMA | Alternate AMA | Comments |
| Early onset VAP | Streptococcus pneumoniae, Haemophilus influenzae, MRSA, E. coli, Klebsiella pneumoniae, Enterobacter spp, Proteus spp, Serratia marscecens | 2nd or 3rd generation cephalosporin eg. Ceftriaxone | BL-BLI eg. Ampicillin+sulbactam 3 gm every 8 hourly Or Ertapenem 1 gm daily | Fluoroquinolon e eg. Levofloxacin or, Moxifloxacin should be avoided due to high prevalence of tuberculosis |
| Late onset VAP or with risk factors for MDR | As in Early VAP, Pseudomonas spp, Klebsiella pneumoniae, E. coli, Acinetobacter spp, MRSA | BL-BLI Piperacillin+tazobactam 4.5 gm 6 hrly or Cefoperazonesulbactam 3 gm 12 hrly, OR Antipseudomonal Carbapenem PLUS Aminoglycoside eg.amikacin 20 mg/kg/day, gentamicin 7 mg/kg/day, tobramycin 7 mg/kg/day Or Antipseudomonal fluoroquinolone e.g. Ciprofloxacin 400 mg 8 hrly, levofloxacin 750 mg daily PLUS Coverage for MRSA e.g. Vancomycin 15 mg/kg every 12 hrs or Linezolid 600 mg 12 hrly | Colistin PLUS Antipseudomonal Carbapenem Or Cefoperazonesulbactam PLUS Coverage for MRSA eg. Vancomycin 15 mg/kg every 12 hrs or Linezolid 600 mg 12 hrly | For pseudomonas double coverage is recommended BL-BLI first choice if sensitivity is >70% For ESBL producing gram- negative strains, carbapenems are appropriate drugs Colistin where carbapenem resistance is high (>70%) |
| Urosepsis | E. coli, Pseudomonas spp, Enterococcus spp., Klebsiella spp., Proteus spp., Anaerobes Candidia spp | BL-BLI or Meropenem or Imipenemcilastatin. Fluconazole if Candida spp. isolated | Colistin with Meropenem; | In pyelonephritis with sepsis, Echiocandins may be considered if Candida species are likely to be fluconzole resistant |
| Intra-abdominal sepsis | E. coli, Pseudomonas spp, Enterococcus spp., Acinetobacter spp, Klebsiella spp., Proteus spp. Candida spp | BL-BLI or Meropenem or Imipenem- cilastatin OR | Colistin with Meropenem; | Source control vital Vancomycin or Teicoplanin if Enterococcus spp isolated Fluconazole or Echinocandins if Candida spp isolated. Echinocandins if prior history of azole exposure or if Candida species are likely to be fluconzole resistant |
| Catheter related blood-stream infection | Gram –negative pathogens
aeruginosa Gram-positive pathogens
aureus, including methicillin-resistant strains Fungi
| Carbapenem, or BL-BLI, with or without an aminoglycoside Vancomycin in settings of high MRSA prevalence; Echinocandin or fluconazole if fungal infection suspected | Add colistin for Gramnegative cover where carbapenem resistance rates are high | Where MRSA isolates have vancomycin MI less than equal to 2 mg/mL, daptomycin, should be used |
| Invasive candidiasis | C. albicans C. tropicalis C. glabrata C. parapsilosis C. krusei | Echinocandin (caspofungin: loading dose 70 mg, then 50 mg daily; micafungin: 100 mg daily; anidulafungin: loading dose 200 mg, then 100 mg daily) as initial therapy | Fluconazole, intravenous or oral, 800 mg (12 mg/kg) loading dose, then 400 mg (6 mg/kg) daily as initial therapy in selected patients, including those who are not critically ill and who are considered unlikely to have a fluconazoleresistant Candida species Lipid formulation amphotericin B (AmB) (3–5 mg/kg daily) if there is intolerance, limited availability, or resistance to other antifungal agents OR Amphotericin deoxycholate (in patients with normal renal function) | Transition from an echinocandin to fluconazole (usually within 5–7 days) is recommended for patients who are clinically stable, have isolates that are susceptible to fluconazole (eg, C. albicans), and have negative repeat blood cultures following initiation of antifungal |
| Febrile neutropenia | Gram-negative pathogens
| Ccarbapenem (meropenem or imipenem-cilastatin), or piperacillin-tazobactam or cefaperazone-sulbactam PLUS aminoglycosides or fluoroquinolones (if judged necessary) PLUS Vancomycin if suspected catheter-related infection, skin and soft-tissue infection, pneumonia, or hemodynamic instability PLUS Initial Antifungal therapy: Echinocandin (In high-risk patients who have persistent fever after 4–7 days ofa broad-spectrum antibacterial regimen and no identified fever source) |
| Modify treatment depending on clinical condition of patient and culture and sensitivity reports |
Note : (1)Antibiotic therapy must be guided by local susceptibility patterns. First line empirical treatment for Gram-negative organisms could be BL-BLI if local susceptibility is 70% and above. For Pseudomonas, Klebsiella and Acinetobacter,use Meropenem or Imipenem if local susceptibility is 70% and above and a combination of Colistin and Meropenem if carbapenem resistance is high (> 70%)
(2)In general, for settings with a higher incidence of ESBL producing organisms, BL-BLI combinations may be used for less ill patients, and carbapenems for patients with greater severity of illness
BL-BLI: beta lactam plus B-lactamase inhibitor: eg; Piperacillin-Tazobactam, Cefaperazone-Sulbactam
Choice of empirical therapy
- The initial management of infection requires forming a probable diagnosis, obtaining cultures, and initiating appropriate and timely empirical antimicrobial therapy and source control (i.e., draining pus, if appropriate)
- Because patients with severe sepsis or septic shock have little margin for error in the choice of therapy, the initial selection of antimicrobial therapy should be broad enough to cover all likely pathogens (bacterial and/or fungal or viral) and that penetrate in adequate concentrations into the tissues presumed to be the source of sepsis.
- Administration of effective intravenous antimicrobials should occur within the first hour of recognition of septic shock and severe sepsis without septic shock.
- Antiviral therapy initiated as early as possible in patients with severe sepsis or septic shock of viral origin.
The choice of empirical therapy depends on:
- the suspected site of infection
- the clinical syndrome
- the setting in which the infection developed (i.e., home, nursing home, or hospital
- medical history
- Epidemiology, susceptibility patterns of bacteria in the hospital and ICU, local microbial-susceptibility patterns, resistance potential
- Prior antibiotic therapy(previous 3 months)
- Immunological competence of patient
- Severity of underlying illness
- Microbes that previously have been documented to colonize or infect the patient. P
- harmacokinetics of the chosen antimicrobial agent
- Drug allergies / toxicities
- Cost
De-escalation
- As soon as the causative pathogen has been identified, de-escalation should be performed by selecting the most appropriate antimicrobial agent that covers the pathogen and is safe and cost-effective.
- The antimicrobial regimen should be reassessed daily for potential de-escalation to prevent the development of resistance, to reduce toxicity, to reduce costs and to reduce the likelihood that the patient will develop superinfection with other pathogenic or resistant organisms, such as Candida species, Clostridium difficile, or vancomycin-resistant Enterococcus faecium.
- Use of low procalcitonin levels or similar biomarkers can assist the clinician in the discontinuation of empiric antibiotics in patients who appeared septic, but have no subsequent evidence of infection
Tropical Infections
Tropical diseases are diseases that are prevalent in, or unique to tropical and subtropical regions. These commonly include dengue hemorrhagic fever, rickettsial infections/scrub typhus, Malaria (usually falciparum), typhoid, and leptospirosis; Bacterial sepsis and viral infections. It was recognized that sometimes the patients may have dual or triple infections and can present with atypical manifestations
There can be no uniform guidelines for empiric therapy but trends of tropical infections should guide the treating physician. The idea is to hit wide and hit early with intention to deescalate once the definitive diagnosis is established.
A syndromic approach to tropical infections can guide the intensivists regarding the commonest etiologies, investigative modalities and help them to choose early empiric therapy. For ease of diagnosis these infections can be divided into 5 major syndromes:
Undifferentiated fever
Malaria (P. falciparum), scrub typhus, leptospirosis, typhoid, dengue fever and other viral illness
Fever with rash/ thrombocytopenia
Bacterial infections, dengue hemorrhagic fever, rickettsial infections/scrub typhus, meningococcal infection, Malaria (usually falciparum), leptospirosis, typhoid, CrimeanCongo hemorrhagic fever and other viral fevers
Fever with ARDS
Falciparum malaria, H1N1 influeza, leptospirosis, hantavirus infection, scrub typhus, Melioidosis, Tuberculosis, severe pneumonias due to legionella and pneumococci, Diffuse alveolar hemorrhage
Febrile encephalopathy
Bacterial infections, Herpes simplex virus encephalitis, Japanese B encephalitis, cerebral malaria, typhoid encephalopathy, fulminant hepatic failure due to viral hepatitis
Fever with multi-organ dysfunction
Bacterial sepsis, Falciparum malaria, Leptospirosis, Scrub typhus, Dengue, Hepatitis A or E with fulminant hepatic failure and hepato-renal syndrome, Hanta virus, Hemophagocytosis and Macrophage activation syndrome
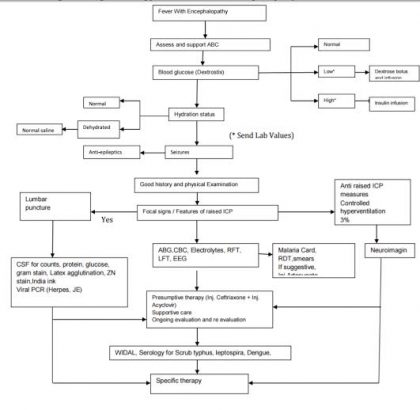
Figure -2 : Algorithmic approach to Fever With Encephalopathy

Disclaimer: This site is primarily intended for healthcare professionals. Any content/information on this website does not replace the advice of medical and/or health professionals and should not be construed as medical/diagnostic advice/endorsement or prescription. Use of this site is subject to our terms of use, privacy policy, advertisement policy. © 2020 Minerva Medical Treatment Pvt Ltd