- Home
- Editorial
- News
- Practice Guidelines
- Anesthesiology Guidelines
- Cancer Guidelines
- Cardiac Sciences Guidelines
- Critical Care Guidelines
- Dentistry Guidelines
- Dermatology Guidelines
- Diabetes and Endo Guidelines
- Diagnostics Guidelines
- ENT Guidelines
- Featured Practice Guidelines
- Gastroenterology Guidelines
- Geriatrics Guidelines
- Medicine Guidelines
- Nephrology Guidelines
- Neurosciences Guidelines
- Obs and Gynae Guidelines
- Ophthalmology Guidelines
- Orthopaedics Guidelines
- Paediatrics Guidelines
- Psychiatry Guidelines
- Pulmonology Guidelines
- Radiology Guidelines
- Surgery Guidelines
- Urology Guidelines
Antimicrobial Guidelines for prophylaxis and treatment of Infections in Bone Marrow Transplant setting
A bone marrow transplant is a procedure to replace damaged or destroyed bone marrow with healthy bone marrow stem cells. Bone marrow is the soft, fatty tissue inside your bones. The bone marrow produces blood cells. Stem cells are immature cells in the bone marrow that give rise to all of your different blood cells.
Indian Council of Medical Research, Department of Health Research has issued the Antimicrobial Guidelines for prophylaxis and treatment of Infections in Bone Marrow Transplant setting. Following are the major recommendations :
Preamble and Case definitions
- Allogeneic transplant : A transplant that uses stem cells taken from a donor (rather than using the recipient's own stem cells).
- Antimicrobial prophylaxis : It refers to the administration of antimicrobial agent (antibiotic, antiviral, anti-fungal or anti-parasitic agent) to the patient usually in lower dose for prevention of infection. It may be administered before (e.g. for surgical intervention), during or after a procedure (e.g. bone marrow transplantation).
- Autologous transplant : A transplant that uses stem cells taken previously from the patient (rather than stem cells from a donor).
- Bone marrow harvesting : A surgical procedure in which doctors insert long needles through the skin to withdraw bone marrow from the crests of the pelvic bones. Donors receive general or spinal anesthesia for the procedure.
- CMV surveillance : Early detection of CMV infection or disease by monitoring of CMV viral load at regular intervals in high risk patients (solid organ transplant and allogeneic bone marrow transplant recipients) for initiation of pre-emptive therapy. Although previously done by CMV cultures and detection of pp65 antigenemia the current technology is to detect CMV viral load by quantitative real time PCR according to standard WHO guidelines for reporting of Quantitative CMV PCR.
- Conditioning : This is chemotherapy, total body irradiation, or both given before a bone marrow transplant. High-dose conditioning is intended to do one of three things:
- Eliminate malignant cells in people with cancer
- Disable the immune system in people with an autoimmune disease
- Destroy the bone marrow in people with other marrow-related diseases
High-dose conditioning leaves patients without an immune system or the ability to form new blood cells.
Alternatively, some patients (typically those who are older or have additional health problems) get reduced-dose conditioning—designed to weaken, but not destroy, their bone marrow and immune system so their body can more readily accept the donor's stem cells.
- Engraftment: Stage in which the bone marrow (or stem cells) of the donor have been taken up by the recipient.
- Haploidentical transplant: A transplant using stem cells from a donor whose HLA type is a half-match for the recipient. This may be an option for people who need a transplant but have not been able to find a more closely matched donor.
- MUD transplant: The genetically matched marrow or stem cells are from an unrelated donor. Unrelated donors are found through bone marrow registries.
- Neutropenic fever: Neutropenia with an absolute neutrophil count (ANC) of <500 cells/mm3 along with single oral temperature measurement of >38.3°C (101°F) or a temperature of >38.0°C (100.4°F) sustained over a 1-hour period.
- Pre-emptive therapy: It is the initiation of specific antimicrobial therapy based on an early diagnostic test with the aim of preventing morbidity due to established disease. For example rising values of a quantitative CMV PCR at weekly intervals precedes establishment of disease and is an indication for "Pre-emptive therapy" with an appropriate antiviral drug. At this time the clinical manifestations of CMV disease are not clinically manifest.
- Stem cell mobilization and collection: Stem cell mobilization means receiving medicine that causes stem cells to leave the tissues they normally occupy and to circulate in the bloodstream. Typically it takes a few days after receiving the medicine for the stem cells to mobilize. Then the stem cells are collected using a machine similar to those used for blood donation at blood banks. A catheter (tube) is placed in a donor's large vein so blood can flow out of the body and into the machine, which separates the stem cells from the blood and returns the blood through another catheter. Collection typically takes a few hours, and donors leave the same day.
- Surveillance culture: Surveillance cultures are cultures obtained from clinical specimens to determine the spectrum of antimicrobial flora and associated resistance patterns in the absence of an obvious clinical infection. In a high risk patient with breach of skin or mucosal barriers there is a possibility that these colonizing microbes would become pathogenic. For example stool surveillance cultures to determine which of these multi-drug resistant organisms (ESBL, AmpC, carbapenemase producers, vancomycin resistant enterococci) are likely to become pathogenic due to gut translocation in a bone marrow transplant recipient.
- Total body irradiation (TBI): TBI is a radiation treatment to the entire body. It is used to destroy cancer cells and bone marrow cells in preparation for a bone marrow transplant.
Table 1 Common pathogens:
| Clinical condition | Common pathogens |
| Community acquired pneumonia | Streptococcus pneumoniae, Haemophilus influenzae, atypical agents (Mycoplasma, Chlamydia, Legionella), respiratory viruses (influenza A, influenza B, RSV, parainfluenza, rhinovirus, human metapneumovirus) |
| Hospital acquired pneumonia | E. coli, Klebsiellla, Pseudomonas, Acinetobacter, Staphylococcus aureus (methicillin resistant and methicillin sensitive) |
| Blood stream infection | E. coli, Klebsiella, Pseudomonas, Acinetobacter, Staphylococcus aureus (methicillin resistant and methicillin sensitive), Staphylococcus epidermidis, Enterococcus species, Candida (albicans and non-albicans species) |
| Intravenous catheter associated infection | Staphylococcus aureus, Staphylococcus epidermidis, Coliforms and non-fermentative Gram negative bacilli |
| Skin and soft tissue infection | Staphylococcus aureus, Streptococcus species, coliforms and nonfermentative Gram Negative Bacilli (in compromised host), candida, aspergillus and zygomycetes |
| Intra-abdominal infection | Coliforms and non-fermentative Gram Negative Bacilli, Anaerobes, Enterococcus species |
| Antibiotic associated diarrhoea | Clostridium difficile |
| Tuberculosis | Mycobacterium tuberculosis |
Figure:1 Phases of opportunistic infections among allogeneic HCT recipients Abbreviations: EBV, Epstein-Barr virus; HHV6, human herpesvirus 6;PTLD, post transplant lympho-proliferative disease
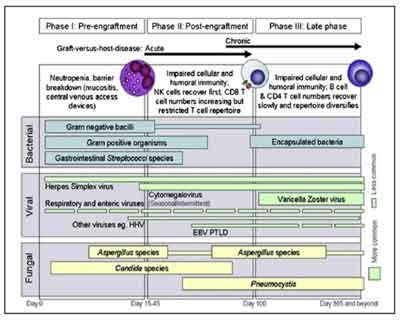 Image source: : Tomblyn M et al. Guidelines for preventing infectious complications among
Image source: : Tomblyn M et al. Guidelines for preventing infectious complications amonghematopoietic cell transplantation recipients: a global perspective. Biol Blood Marrow
Transplant. 2009 Oct;15(10):1143-238.
Table 2 Investigations:
| Diagnostic sector | Investigations relevant for management of infections and its complications |
| Hematology | Complete Blood Count, Differential count, prothrombin time, APTT, INR |
| Biochemistry | Urea, creatinine, electrolytes (sodium, potassium, calcium, magnesium, phosphorus), glucose, liver function tests (bilirubin- total, direct, indirect, SGOT, SGPT, alkaline phosphatase), C-reactive protein, procalcitonin |
| Microbiology | Blood stream infection: blood culture (from peripheral vein as well as through a central line if present) Urinary tract infection: urine microscopy and culture sensitivity (midstream or catheterized urine) Gastroenteritis: Stool microscopy for ova cysts parasites and culture sensitivity Antibiotic associated diarrhea: Clostridium difficile toxin detection Upper respiratory tract infection: Nose and throat swab or nasopharyngeal aspirate for respiratory virus PCR (influenza A and B, parainfluenza viruses, respiratory syncitial viruses, rhinovirus, human metapneumovirus), throat swab for bacterial culture Lower respiratory tract infection (pneumonia, hospital acquired pneumonia, ventilator associated pneumonia): Bacterial and fungal culture of sputum, broncho-alveolar lavage, endotracheal aspirate Skin and soft tissue infection including surgical site infection: Pus swab, aspirate or tissue for bacterial+/- fungal culture CMV viremia: CMV viral load determination by quantitative real time PCR CMV disease (colitis/pneumonia/hepatitis/encephalitis): CMV viral load determination by quantitative real time PCR Detection of multidrug resistant bacteria: MRSA screening: nose swab surveillance culture for MRSA Stool surveillance culture: for detection of ESBL, AmpC, carbapenemase producers and vancomycin resistant enterococci Serological screening before transplantation of donor and recipient: HIV, HBsAg, anti-HCV, HTLV, CMV IgG Tuberculosis : Mycobacterial culture from relevant samples |
| Imaging | Chest X-Ray, ultra sonography, CT scan, MRI, PET scan |
a. Resistance pattern of common pathogens:
Table 3 . Staphylococcus aureus ICMR AMR Data 2014
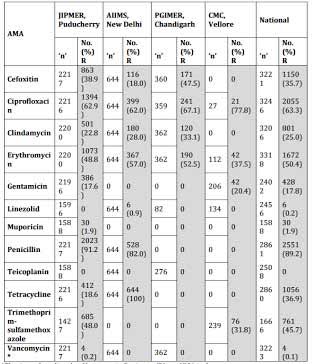
*The 4 numbers listed as Vancomycin Resistant (R) are VISA isolates.
No VRSA was isolated during the year 2014 at JIPMER.
Cefoxitin : Surrogate marker for Methicillin.
Table 4. Enterococcus faecium ICMR AMR Data 2014
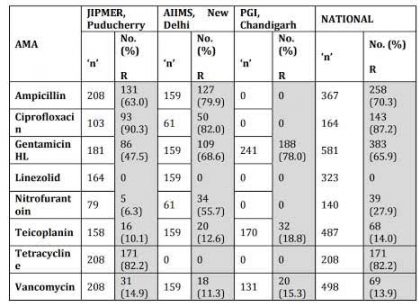
Table5 . Enterobacteriaceae isolates from blood. ICMR AMR data 2014
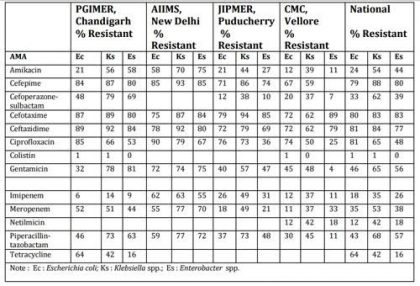
Table 6. Pseudomonas aeruginosa ICMR AMR Data 2014
| AMA | PGIMER, Chandigarh 'n' 75 R (%) | AIIMS, New Delhi 'n' 102 R (%) | JIPMER, Puducherry 'n' 113 R (%) | CMC, Vellore 'n' 84 R (%) | National 'n' 374 R % |
| Amikacin | 27 | 49 | 38 | 21 | 35 |
| Aztreonam | 62 | 55 | 30 | 48 | |
| Cefepime | 52 | 57 | 20 | 41 | |
| Cefoperazone-sulbactam | 39 | 41 | 30 | 38 | |
| Ceftazidime | 64 | 51 | 51 | 23 | 47 |
| Colistin | 34 | 2 | 10 | ||
| Imipenem | 17 | 54 | 48 | 25 | 37 |
| Levofloxacin | 44 | 42 | 23 | 36 | |
| Meropenem | 74 | 41 | 23 | 47 | |
| Netilmicin | 66 | 45 | 22 | 45 | |
| Piperacillin-tazobactam | 44 | 67 | 25 | 46 | |
| Tobramycin | 56 | 43 | 18 | 33 |
Table 7. Acinetobacter baumannii ICMR AMR Data 2014
| AMA | PGIMER, Chandigarh 'n' 209 R (%) | AIIMS, New Delhi 'n' 143 R (%) | JIPMER, Puducherry 'n' 157 R (%) | CMC, Vellore 'n' 90 R (%) | National 'n' 599 R % |
| Amikacin | 77 | 83 | 59 | 84 | 75 |
| Aztreonam | 87 | 93 | 84 | 87 | |
| Cefepime | 98 | 86 | 75 | 61 | 81 |
| Cefoperazone-sulbactam | 89 | 23 | 22 | 47 | 57 |
| Ceftazidime | 99 | 86 | 68 | 68 | 84 |
| Colistin | 1 | 64 | 22 | 22 | |
| Imipenem | 52 | 83 | 62 | 64 | 63 |
| Levofloxacin | 86 | 68 | 60 | 73 | |
| Meropenem | 50 | 86 | 59 | 61 | 62 |
| Netilmicin | 79 | 56 | 69 | ||
| Piperacillin-tazobactam | 73 | 86 | 71 | 83 | |
| Tetracycline | 61 | 52 | 55 | ||
| Tobramycin | 54 | 64 | 58 | 58 | |
| Trimethoprim-sulphamethoxazol e | 46 | 63 | 55 |
Table 8 . Candida spp. isolated at PGIMER, Chandigarh ICMR AMR Data 2014.
| AMA | C tropicalis 'n' 101; % R | C krusei 'n' 98; % R | C albicans 'n' 50; % R | C pelliculosa 'n' 35; % R |
| Amphotericin B | 1 | 5 | 0 | 0 |
| Fluconazole | 4 | 0 | 8 | 0 |
| Voriconazole | 1 | 3 | 8 | 0 |
| Itraconazole | 8 | 4 | 2 | 0 |
| Posaconazole | 1 | 0 | 0 | 0 |
| Caspofungin | 2 | 8 | 0 | 0 |
| Anidulafungin | 1 | 7 | 2 | 0 |
| Micafungin | 0 | 6 | 2 | 6 |
II. Table 9 Antimicrobial Agents (AMA) regime (based on ICMR/Indian resistance data) in the Bone Marrow Transplant setting:
| Clinical Condition | Common Pathogens | Empirical Antimicrobial Agents | Alternate Antimicrobial Agents | Comments |
| Neutropenic fever/ sepsis | Escherichia coli, Klebsiella pneumoniae, Pseudomonas aeruginosa, Acinetobacter species, Staphylococcus aureus, Coagulase Negative Staphylococci, Enterococcus species, Candida species | Piperacillin-tazobactam + amikacin | Second line: Meropenem± Teicoplanin/Vancomycin. Third line or patient in septic shock: Meropenem+ Colistin± Teicoplanin/Vancomycin+ caspofungin | Continue broad-spectrum antibiotics until the patient has been afebrile for at least 2 days and the neutrophil count is >500 cells/mm3 on at least one occasion. Gram positive cover with Teicoplanin or Vancomycin to be discontinued once cultures are negative. |
| Community acquired pneumonia | Streptococcus pneumoniae, Haemophilus influenzae, atypical agents (Mycoplasma, Chlamydia, Legionella), respiratory viruses (influenza A, influenza B, RSV, parainfluenza, rhinovirus, human metapneumovirus) | Piperacillin-tazobactam+ clarithromycin or azithromycin | Meropenem+ clarithromycin or azithromycin+ Teicoplanin/Vancomycin | If viral infection is suspected send respiratory sample (nose and throat swab/BAL/endotracheal secretion) for respiratory viral PCR and consider early empirical use of oseltamivir to be discontinued once influenza PCR is negative |
| Hospital acquired pneumonia | E. coli, Klebsiella, Pseudomonas, Acinetobacter, Staphylococcus aureus (methicillin resistant and methicillin sensitive) | Piperacillin-tazobactam + amikacin | Meropenem+ Teicoplanin/Vancomycin | Consider use of colistin with meropenem and Teicoplanin/Vancomycin in case of severe infection requiring respiratory support (ventilation) and this may be discontinued once cultures are negative. Also consider use of aerosolized colistin as an adjunct to intravenous antibiotics in the treatment of multi-drug resistant pathogens Where toxicity is a concern. |
| Blood stream infection | E. coli, Klebsiella, Pseudomonas, Acinetobacter, Staphylococcus aureus (methicillin resistant and methicillin sensitive), Staphylococcus epidermidis, Enterococcus species, Candida (albicans and non-albicans species) | Piperacillintazobactam + amikacin Or Cefoperazone-Sulbactam+ amikacin | Second line: Meropenem± Teicoplanin/Vancomycin Third line or patient in septic shock: Meropenem+ Colistin± Teicoplanin/Vancomycin + caspofungin | Duration of treatment depends on the actual or the suspected source of blood stream infection |
| Intravenous catheter associated infection | Staphylococcus aureus, Staphylococcus epidermidis, Coliforms and non-fermentative Gram negative bacilli | Piperacillin-Tazobactam+ Vancomycin | Meropenem+ Teicoplanin/Vancomycin | Consider the use colistin or antifungal agents based on specific clinical/laboratory diagnosis |
| Skin and soft tissue infection | Staphylococcus aureus, Streptococcus species, coliforms and non-fermentative Gram Negative Bacilli (in compromised host), candida, zygomycetes | Piperacillin- Tazobactam + Teicoplanin/ Vancomycin | Meropenem+ Teicoplanin/Vancomycin + Clindamycin | Consider the use of anti-fungal agents based on specific clinical/laboratory diagnosis. For MRSA coverage consider use of Teicoplanin/Vancomycin combination. Consider the use of clindamycin where anti-toxin activity is desired (e.g. necrotizing fasciitis). |
| Intraabdominal infection | Coliforms and non-fermentative Gram Negative Bacilli, Anaerobes, Enterococcus species, Candida | Piperacillin-Tazobactam+ Amikacin ± metronidazole | Meropenem+ Teicoplanin/Vancomycin ± metronidazole | Consider the use of anti-fungal agents based on specific clinical/laboratory diagnosis |
| Urinary Tract infection | Coliforms and non-fermentative Gram Negative Bacilli, Enterococcus species, Candida | Piperacillin-Tazobactam+ Amikacin | Meropenem+ Teicoplanin/Vancomycin | Consider the use of anti-fungal agents based on specific clinical/laboratory diagnosis |
| Antibiotic associated diarrhoea | Clostridium difficile | Metronidazole | Oral Vancomycin | Oral Vancomycin may be used as first line in severe infections |
| Invasive pulmonary aspergillosis | Aspergillus flavus, Aspergillus fumigatus, Aspergillus nidulans, Aspergillus niger, Aspergillus terreus | Voriconazole | Amphotericin B (preferably liposomal, otherwise conventional), with or without Caspofungin | 6-12 weeks. Treatment should be continued until lesions have resolved or clinically stable |
| Mucormycosis | Apophysomyces, Basidiobolus, Conidiobolus, Cunninghamella, Mortierella, Mucor, Lichtheimia (Absidia), Rhizomucor, Rhizopus, Saksenaea, Syncephalestrum | Liposomal Amphotericin B with Surgical debridement (wherever feasible) | Caspofungin may be considered along with liposomal amphotericin B | Surgical debridement as far as possible. Antifungal therapy for mucormycosis should be continued until: there is resolution of clinical signs and symptoms of infection, there is resolution or stabilization of residual radiographic signs , there is resolution of underlying immunosuppression. Adjunctive therapies may be tried in case of non response. |
| Herpes simplex | Herpes Simplex Virus Type 1 and Type 2 (HSV1, HSV2) | Acyclovir or Valacyclovir | Foscarnet | Duration of therapy depends on organ involvement. |
| Varicella or disseminated zoster or localized zoster | Varicella Zoster Virus | Acyclovir or Valacyclovir | Foscarnet | Duration of therapy depends on organ involvement Requires a minimum of 7-10days of treatment with acyclovir/ valacyclovir in case of cutaneous involvement |
| CMV reactivation or disease (colitis, pneumonitis, hepatitis, retinitis, encephalitis) | Cytomegalovirus | Ganciclovir or Valgancyclovir | Foscarnet or Cidofovir | Treat till resolution of clinical symptoms and signs or resolution of viremia (2 negative viral load reports) |
| Pneumocystis jirovecii pneumonia | Pneumocystis jirovecii | Co-trimoxazole (high dose) | Clindamycin+ Primaquine | Duration of therapy:21 days |
III. Foot Notes:
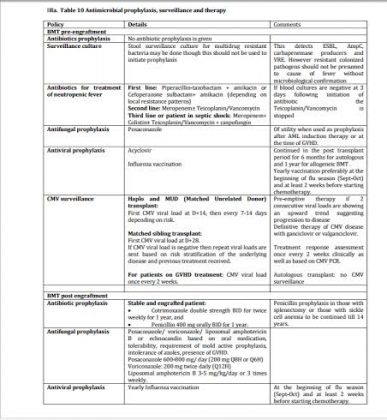
IIIa. Table 10 Antimicrobial prophylaxis, surveillance and therapy
IIIb. Table 11 Standard adult doses of antimicrobial agents
| Antimicrobial agent | Standard adult dose |
| Acyclovir | Prophylaxis: 400 mg Q12H (BID) for a month after BMT and then shift to 800 mg Q12H (BID) for a year. This is to prevent Herpes simplex initially but Herpes Zoster at a later stage Herpes simplex in immunocompromised requiring IV treatment: 5 mg/kg body weight per dose for Q8H (TID). Varicella zoster: 800 mg orally 5 times daily ; In immunocompromised 10 mg/kg body weight Q8H (TID) |
| Amikacin | 15 mg/kg IV or IM Q24H (OD) |
| Amphotericin B (conventional) | 0.5-1 mg/kg body weight IV Q24H (OD) in 5% dextrose over 4 hours after prehydration |
| Amphotericin B (liposomal) | 3-5 mg/kg body weight IV Q24H (OD) |
| Anidulafungin | 200 mg loading dose on Day 1 followed by 100 mg on Day 2 IV Q24H (OD) |
| Azithromycin | 500 mg IV Q24H (OD) |
| Caspofungin | 70 mg IV as loading dose followed by 50 mg IV Q24H (OD) from next day |
| Ciprofloxacin | 400 mg IV Q8H (TID) or 750 mg PO Q12H (BID) |
| Clindamycin | 300-600 mg IV Q6H (QID) |
| Colistin | 9 MU loading followed 12 hours later by 4.5 MU IV Q12H (BID); |
| Co-trimoxazole | Treatment of Pneumocytis jiroveci: 20 mg/kg of trimethoprim with 100 mg/kg of sulphamethoxazole in 2-4 divided doses; Treatment of Nocardiosis: DS tab (960 mg) 3-4 times daily for 3-6 months. Cerebral disease is to be treated longer |
| Fluconazole | For invasive candidiasis: 800 mg loading dose followed 24 hours later by 400 mg Q24H (OD); For other indications please look up appropriate guidelines |
| Ganciclovir | 5 mg/kg body weight Q12H (BID) (10 mg/kg per day) |
| Gentamicin | 5 mg/kg IV Q24H (OD) |
| Imipenem | 500 mg IV Q6H (QID) |
| Levofloxacin | 500 mg-750mg IV Q24H (OD) |
| Meropenem | 1 gm IV Q8H (TID) |
| Metronidazole | 500 mg IV Q8H (TID) |
| Micafungin | 100 mg IV OD |
| Oseltamivir | 75 mg orally Q12H (BID) |
| Piperacillin-tazobactam | 4.5 gm IV Q8H (TID) |
| Posaconazole | Prophylaxis: 200 mg Q8H (TID) or Q6H (QID) orally with fatty food |
| Teicoplanin | 400 mg IV Q12H (BID) for day 1 then 400 mg IV Q24H (OD) |
| Tigecycline | Initial dose of 100 mg followed by 50 mg IV Q12H (BID) |
| Valacyclovir | Varicella zoster: 1000 mg orally Q8H (TID) Herpes simplex: 1000 mg orally Q12H (BID) |
| Valganciclovir | 900 mg orally Q12H (BID) |
| Vancomycin | 1 gm IV Q12H (BID) |
| Voriconazole | 400 mg IV Q12H (BID) on day 1 followed by 200 mg IV Q12H (BID) from next day |
Note:
- Dose of the antimicrobial agent depends on the age, indication, site and severity of infection, body weight, creatinine clearance/ renal and hepatic function
- Modify dose and duration as required based on age, indication, site and severity of infection, body weight, creatinine clearance/ renal and hepatic function
- Doses mentioned in the above table reflect those used in adults without renal or hepatic impairment and serious infections. Wherever applicable prophylactic dosing has been indicated
- A higher dose of colistin has been recommended for critically ill patients [9 MU IV as the loading dose followed 24 hours later by 4.5 MU IV BID];
- OD- once daily (Q24H); BID- twice daily (Q12h); TID- three times daily (Q8h); QID- four times daily (Q6h)
- For extensively drug resistant organisms such as colistin-resistant Klebsiella pneumoniae please refer to antibiotic susceptibility test results and specific references
IIIc. Table 12 Duration of Antimicrobial Therapy
| Category | Duration |
| Fever of unidentified etiology | Continue broad-spectrum antibiotics until the patient has been afebrile for at least 2 days and the neutrophil count is >500 cells/mm3 on at least one occasion. |
| Documented infections | The duration of antibiotic therapy should be appropriate for effective eradication of the identified infection. |
| Bacterial bloodstream infections, soft-tissue infections, and pneumonia | 10–14 days of appropriate antibiotic therapy. Antibiotic treatment may therefore extend beyond resolution of fever and neutropenia. |
| Deep tissue infection, endocarditis and septic thrombosis, complicated central line associated blood stream infection (CLABSI) persistent bacteremia or fungemia occurring >72 hour after catheter removal | Treatment duration depends on the organism and extent of involvement. Please involve an ID physician to determine appropriate antibiotic and duration of therapy. |
| CLABSI caused by Staphylococcus. aureus, Pseudomonas aeruginosa, fungi, or mycobacteria | Systemic antimicrobial therapy for at least 14 days along with catheter removal if there are no metastatic complications |
| Candidemia without obvious metastatic complications | 2 weeks after documented clearance of Candida from the bloodstream and resolution of symptoms attributable to candidemia and resolution of neutropenia |
| Deep seated infection with candida (endophthalmitis, septic arthritis, infection of prosthetic devices, endocarditis etc) | Prolonged treatment (minimum of 4–6 weeks) is recommended. For candida endocarditis valve replacement is recommended, and treatment should continue for at least 6 weeks after valve replacement and should continue for a longer duration in patients with perivalvular abscesses and other complications |
| Antifungal prophylaxis | For the duration of the neutropenia |
| Invasive pulmonary aspergillosis | 12 weeks. Treatment should be continued until lesions have resolved or are clinically stable |
| Mucormycosis | Antifungal therapy for mucormycosis should be continued until all of the following objectives are attained: (1) there is resolution of clinical signs and symptoms of infection, (2) there is resolution or stabilization of residual radiographic signs of disease on serial imaging, and (3) there is resolution of underlying immunosuppression. |
| Influenza | 5 days of treatment with oseltamivir |
| Herpes simplex | 5-10 days |
| Varicella zoster | 7 days of treatment with acyclovir/ valacyclovir |
| CMV | As directed by viral load measurements |
| Pneumocystis jirovecii pneumonia | 14-21 day |
IIId. De-escalation
If the patient is afebrile, signs of severe sepsis / septic shock and neutropenia have resolved, plan for targeted therapy based on culture and susceptibility reports.
You can read the full Guideline by clicking on the link :
Dr Soumya Swaminathan, Director General, Indian Council of Medical Research Secretary, Department of Health Research

Disclaimer: This site is primarily intended for healthcare professionals. Any content/information on this website does not replace the advice of medical and/or health professionals and should not be construed as medical/diagnostic advice/endorsement or prescription. Use of this site is subject to our terms of use, privacy policy, advertisement policy. © 2020 Minerva Medical Treatment Pvt Ltd