- Home
- Editorial
- News
- Practice Guidelines
- Anesthesiology Guidelines
- Cancer Guidelines
- Cardiac Sciences Guidelines
- Critical Care Guidelines
- Dentistry Guidelines
- Dermatology Guidelines
- Diabetes and Endo Guidelines
- Diagnostics Guidelines
- ENT Guidelines
- Featured Practice Guidelines
- Gastroenterology Guidelines
- Geriatrics Guidelines
- Medicine Guidelines
- Nephrology Guidelines
- Neurosciences Guidelines
- Obs and Gynae Guidelines
- Ophthalmology Guidelines
- Orthopaedics Guidelines
- Paediatrics Guidelines
- Psychiatry Guidelines
- Pulmonology Guidelines
- Radiology Guidelines
- Surgery Guidelines
- Urology Guidelines
Antibiotic-releasing polymer to help eradicate joint implant infection
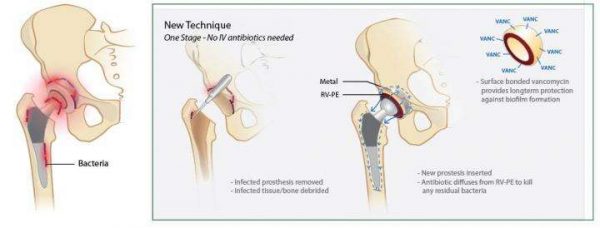
A team of Massachusetts General Hospital (MGH) investigators has developed an antibiotic-releasing polymer that may greatly simplify the treatment of prosthetic joint infection. In their recent report published in Nature Biomedical Engineering, the researchers describe how implants made from this material successfully eliminated two types of prosthetic infection in animal models.
"Currently, most infections involving total joint replacement prostheses require a two-stage surgery, in which the patient's daily activities are largely compromised for four to six months," says Orhun Muratoglu, PhD, director of the Harris Orthopædics Laboratory in the MGH Department of Orthopædic Surgery, a co-author of the report. "Our finding that polyethylene, the most commonly used weight-bearing surface in total joint surgery, can be made to safely and effectively release antibiotics implies that fully weight-bearing implants made with this material could be used to treat infection in a single procedure, reducing both the inconvenience and the risk of complications for patients."
Delivering antibiotics to an infected prosthetic joint is challenging because of the limited supply of blood to the area. As Muratoglu describes, the standard treatment for prosthetic joint infection in the U.S. -- which affects up to 30,000 people each year -- involves removal of the implant and adjacent infected tissues and placement of a temporary spacer made from antibiotic-releasing bone cement that remains within the joint space for at least six weeks and sometimes for as long as six months. During that time, the patient's movement may be significantly restricted, depending on the involved joint. In a second surgery, a new prosthesis is implanted, using antibiotic-releasing bone cement. But patients still can be at risk for recurrent infection, which may lead to the need for permanent joint fusion or amputation and has a 10-15 percent mortality rate.
Antibiotic-releasing bone cement has several limitations. Its ability to release an effective antibiotic dose may be brief, lasting little more than a week, and increasing the antibiotic content reduces the material's durability. In addition, some antibiotics with desirable qualities cannot be incorporated into a bone cement. For the current study, the research team -- including lead author Jeremy Vincentius Suhardi, a Harvard/MIT MD/PhD student, and senior author Ebru Oral, PhD, both of the Harris Lab -- designed and developed an antibiotic-releasing polymer that could be incorporated into the implant itself.
Based on mathematical and statistical models, the material they developed contained antibiotic clusters which were irregularly shaped, making them able to release effective drug doses over extended periods of time without compromising the strength of the material. Implants made from this polymer were tested in animal models of prosthetic joint infection produced either by injecting a Staph. aureus-containing solution into the prosthesis or implanting a titanium rod covered with a Staph. Aureus biofilm, a coating of bacteria that is particularly difficult to treat. In both situations, the antibiotic-releasing polymer successfully eliminated the infection, while implantation of a drug-release bone cement spacer was not effective.
"We used two separate infection models because, when patients present with prosthetic joint infection symptoms, it is not clear what proportion of bacteria may be in a biofilm and what are free floating in solution," says Muratoglu. "The ability of our devices to eradicate all bacteria in the joints in both models strongly suggests they would be successful against both types of periprosthetic infection." A professor of Orthopedic Surgery at Harvard Medical School, Muratoglu notes that, in addition to speeding the recovery of patients and reducing the chance of complications, the elimination of a second surgical procedure should reduce overall costs.
He adds that the successful development of devices like these implants relies on an institutional "ecosystem," involving scientists, engineers and surgeons who can identify a problem, form hypotheses and strategies, and develop and test materials to meet the challenges. The team is now working with the Food and Drug Administration and other regulatory agencies to pursue necessary approvals and develop this material into clinical products.
For more details click on the link : V. J. Suhardi, D. A. Bichara, S. J. J. Kwok, A. A. Freiberg, H. Rubash, H. Malchau, S. H. Yun, O. K. Muratoglu, E. Oral. A fully functional drug-eluting joint implant. Nature Biomedical Engineering, 2017; 1 (6): 0080 DOI: 10.1038/s41551-017-0080

Disclaimer: This site is primarily intended for healthcare professionals. Any content/information on this website does not replace the advice of medical and/or health professionals and should not be construed as medical/diagnostic advice/endorsement or prescription. Use of this site is subject to our terms of use, privacy policy, advertisement policy. © 2020 Minerva Medical Treatment Pvt Ltd