- Home
- Editorial
- News
- Practice Guidelines
- Anesthesiology Guidelines
- Cancer Guidelines
- Cardiac Sciences Guidelines
- Critical Care Guidelines
- Dentistry Guidelines
- Dermatology Guidelines
- Diabetes and Endo Guidelines
- Diagnostics Guidelines
- ENT Guidelines
- Featured Practice Guidelines
- Gastroenterology Guidelines
- Geriatrics Guidelines
- Medicine Guidelines
- Nephrology Guidelines
- Neurosciences Guidelines
- Obs and Gynae Guidelines
- Ophthalmology Guidelines
- Orthopaedics Guidelines
- Paediatrics Guidelines
- Psychiatry Guidelines
- Pulmonology Guidelines
- Radiology Guidelines
- Surgery Guidelines
- Urology Guidelines
All About Sudden cardiac arrest- By Dr Srikant Sharma
Sudden cardiac arrest (SCA) is a condition in which the heart suddenly and unexpectedly stops beating thereby jeopardising the blood stops the flow to the brain and other vital organs and it may lead to if it's not treated within minutes. Dr Srikant Sharma has exhaustively covered all the aspects of Sudden Cardiac Arrest in an exhaustive manner for readers of Specialty Medical Dialogues.
Q1) What is Sudden Cardiac Arrest?
Sudden cardiac arrest (SCA) is defined as “sudden cessation of cardiac activity resulting in the victim becoming unresponsive with no normal breathing and no signs of circulation, within 1 hour of presentation of symptoms”. The condition generally results from an electrical disturbance in the heart that disrupts its ability to function.
In simple terms, if you were to consider the heart to be a house, Sudden Cardiac Arrest is an electricity supply problem whereas a Heart Attack is a plumbing problem; Heart Attack/Myocardial Infarction being tissue death (infarction) of the heart muscle (myocardium) resulting from nil or severely restricted blood flow in
the corresponding coronary artery supplying it.
A Sudden Cardiac Arrest in almost 50% of cases occurs suddenly without any symptoms and if corrective measures aren’t taken rapidly, the condition progresses to Sudden Cardiac Death. (SCD)
Q2) How common is Sudden Cardiac Arrest?
In the USA, Sudden Cardiac Arrest outside hospital occurs in 13/10,000 people per year (326,000 cases) In-hospital cardiac arrest occurs in an additional 209,000. survival rates with treatment being 8%.
In Delhi approximately 25 to 45 people die daily due to SCA.
Based on death certificates, Sudden Cardiac Deaths accounts for about 15% of all deaths in western countries. The lifetime risk is three times greater in men (12.3%) than women (4.2%) based on analysis of the Framingham Heart Study.
However, this gender difference disappears after 85 years of age.
Q3) Am I, as an Indian at a higher risk for SCA?
In India, the incidence of SCD is a grossly understudied area, studies done project an incidence of 39.7/100,000 on mathematical grounds in people of ages over 35
years of age. In comparison to the west, the peak incidence group of SCDs in India was 5-8 yrs younger.
Survival rate in India is less than 1%; major contributory factor to it being ignorance pertaining to CPR specifically in Out-Of-Hospital settings.
Widespread availability of CPR training in general population and availability of Automatic External Defibrillators (AED) can bring about a huge difference in the survival rates undoubtedly.
Q4) What are the risk factors?
Risk factors for Sudden Cardiac Death are similar to those of Coronary Artery Disease. Factors like smoking, lack of physical exercise, hypertension, non-HDL cholesterol, central apple shaped obesity, diabetes mellitus and significant family history.
Smoking increases the risk of SCDs 2-3 folds in ages of 30-59yrs. Also to be noted is that the risk of SCDs in smokers who have quit smoking reaches levels closer to those who have never smoked, provided the period of cessation of smoking has been 10 years or more.
Another significant factor which has found its way into being classified as a major risk factor is Air Pollution especially in cities like Delhi/Beijing amongst others.
Q5) What are the causes of Sudden Cardiac Death?
Theoretically, it can be classified into 4 separate etiological groups:
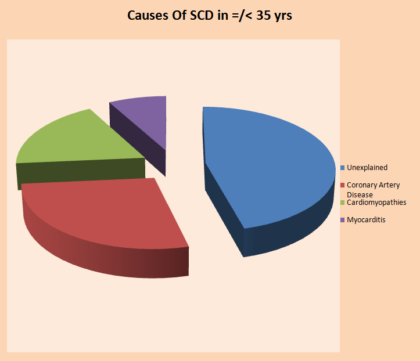
1) Ischaemic Heart Disease
2) Non Ischaemic Heart Disease/ Structural Heart Disease
3) Non Structural Heart Disease
4) Non Cardiac Causes
Q6) What Ischaemic Heart Diseases lead to Sudden Cardiac Arrest/Sudden Cardiac Death?
This etiological group alone contributes to about 70% of all cases.
- Coronary artery disease with myocardial infarction or angina
- Coronary artery embolism
- Non Atherogenic coronary artery diseases – arteritis, dissection, congenital coronary artery anomalies.
- Coronary artery spasm.
Q7) What are the Non Ischaemic Heart Diseases/Structural Heart Diseases leading to Sudden Cardiac Arrest?
10% of all Sudden Cardiac Arrest cases are contributed by this particular group of etiologies.
- Hypertrophic Cardiomyopathy
- Dilated Cardiomyopathy
- Valvular Heart Disease
- Congenital Heart Disease
- Arrhythmogenic Right Ventricular Dysplasia
- Myocarditis
- Acute Pericardial Tamponade
- Acute Myocardial Rupture
- Aortic Dissection
Q8) What is the leading cause of death amongst Non Ischaemic Heart Diseases?
Left Ventricular Hypertrophy is thought to be the leading cause of death from this particular etiological group.
Q9) Does Congestive Heart Failure increase the risk of SCA/SCD?
Congestive Heart Failure increases the risk by 5 times.
Q10) What are the Non Structural Heart Diseases contributing to Sudden Cardiac Arrest / Sudden Cardiac Death?
5-10% is estimated to be the contribution of this etiological group towards Sudden Cardiac Arrests.
- Long QT syndrome (It affects repolarization of the heart after a heartbeat. This results in the affected individual being at a predisposed risk of an irregular heartbeat. Incidence in the west being 1 in 5000 to 7000 newborns and is projected to be the cause for 3000 deaths each year out of 300,000 [cardiac arrests seen by emergency departments.)
- Brugada syndrome (It is a genetic disorder in which the electrical activity within the heart is inherently abnormal; incurable per se, it can be managed with ICD therapy.)
- Catecholaminergic polymorphic ventricular tachycardia (It is an inherited genetic disorder that predisposes those affected to potentially life-threatening abnormal heart rhythms or arrhythmias. The arrhythmias generally occur during exercise or at times of emotional stress; typically presenting as bidirectional ventricular tachycardia or ventricular fibrillation)
Q11) What about Non Cardiac causes?
Sudden Cardiac Arrests due to non-cardiac causes accounts for the remaining 15 to 25%.
- Trauma
- GI Bleeding
- Drowning
- Drug Overdose
- Sudden infant death
- Misc: eg. Stings of certain Jellyfish species.
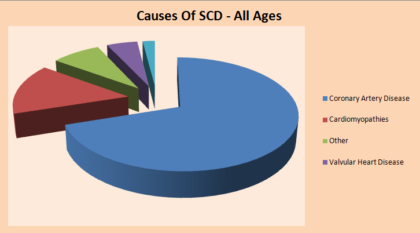
Q12) What is the age wise distribution of causes in Sudden Cardiac Arrest/Sudden Cardiac Death?

Q13) How does a healthcare professional diagnose Sudden Cardiac Arrest?
For ages, the absence of a pulse has been the backbone of diagnosing a Sudden Cardiac Arrest. Nonetheless, studies have shown that rescuers often make a mistake when checking the carotid pulse in an emergency, whether they are healthcare professionals or lay persons. Human errors aside, certain conditions on presentation to the Emergency Department elicit an extremely feeble pulse which may not be felt and hence, misdiagnosed as a Cardiac Arrest. One notorious example being Shock.
Due to the non-specificity in this method of diagnosis, the ERC (European Resuscitation Council) amongst others have played down the importance of using Pulse alone as the primary gold standard diagnostic tool. The Resuscitation Council (UK), in conjunction with the ERC's recommendations and those of the American Heart Association have suggested that the technique should be used only by healthcare professionals with specific knowledge, and even then it should be viewed along with the other pointers of life.
The International Liaison Committee on Resuscitation (ILCOR) recommendations were for healthcare professionals to search for "signs of circulation" and not just the pulse alone. [28] These included gasping, coughing, color-touch, twitching and movement. However, in face of evidence that these guidelines were not completely effective, ILCOR currently advises that sudden cardiac arrest should be diagnosed in all patients who on presentation are unconscious/low Glasgow Coma Scale score and not breathing normally.
Q14) How is Sudden Cardiac Arrest treated upon presentation in the Emergency Department?
- It is treated as per the AHA 2015 guidelines in modern Emergency Medicine Departments.
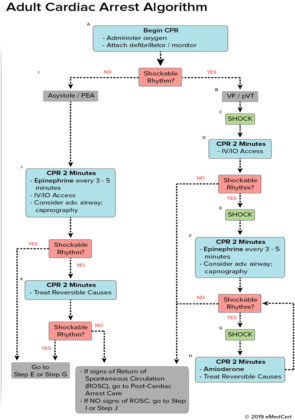
- Abbreviations:
- Vf – Ventricular Fibrillation
- pVT – Pulseless Ventricular Tachyardia
- PEA – Pulseless Electrical Activity
- ROSC – Return of Spontaneous Circulation.
- IO- Intravenous.
- IV-Intravenous
Q15) What entails a good quality CPR?
- Using both hands, place the heel of one over the sternum at the nipple level; push hard (atleast 2 inches = 5cms ) and fast (100-120/min), allowing complete chest recoil.
- Minimize interruptions in compressions.
- Avoid excessive ventilation.
- Rotate compressor every 2 mins, or sooner if fatigues.
- If no advanced airway established, maintain 30:2 compression – ventilation ratio.
- Quantitative waveform capnography – If ETCO2 (expiratory carbon dioxide) <10mmhg, improve CPR quality.
Q16) What are the shock-able rhythms and what is the Shock Energy used for defibrillation?
- Ventricular Fibrillation (V-fib/vF) & Pulseless Ventricular Tachycardia(pVT) are the two shock-able rhythms. Biphasic: Manufacturer recommendation (eg. Initial dose of 120-200 Joules; if unknown then use the maximum available) Second and subsequent doses should be equivalent and higher doses may be considered.
- If Monophasic: 360 Joules.
Q17) Which drugs are part of the treatment algorithm for Sudden Cardiac Arrest and what are their doses?
- Epinephrine: IV/IO dose - 1mg every 3-5 minutes.
- Amiodarone: IV/IO dose – First dose being 300mg bolus; Second dose being 150mg.
Q18) What are the 2018 AHA Review/2018 CoSTR (Consensus on CPR and ECC Science with Treatment Recommendation) inputs which intend to contribute towards updated standardized care and rise in rates of ROSC achieved?
The review considered the use of Amiodarone, Lidocaine, Magnesium and Beta-blockers for antiarrhythmic therapy during and immediately after adult ventricular
fibrillation (VF) and pulseless ventricular tachycardia (pVT) cardiac arrest.
Q19) Can Amiodarone or Lidocaine be considered for VF/pVT that is unresponsive to defibrillation?
As per the 2018 review, Amiodarone or lidocaine may be considered for VF/pVT that is unresponsive to defibrillation (3 shocks).
These drugs may be particularly useful for patients with witnessed arrest, for whom time to drug administration may be shorter (Class IIb, LOE B-R).
Q20) Can Magnesium be routinely used for treating cardiac arrest?
As reviewed in 2018, the routine use of magnesium for cardiac arrest is not recommended in adult patients (Class III: No Benefit, LOE C-LD). Magnesium may be considered for torsades de pointes (ie, polymorphic VT associated with long QT interval) (Class IIb, LOE C-LD).
Q21) What is the take on using Beta blockers within the first hour after ROSC?
As per the 2018 review, there is insufficient evidence to support or refute the routine use of a β-blocker early (within the first hour) after ROSC.
Q22) Has Lidocaine been deemed helpful for routine use?.
As per the 2018 review, there is insufficient evidence to support or refute the routine use of lidocaine early (within the first hour) after ROSC. In the absence of contraindications, the prophylactic use of lidocaine may be considered in specific circumstances (such as during emergency medical services transport), when treatment of recurrent VF/pVT might prove to be challenging.
Q23) How is SCD assessed & investigated?
It is imperative to investigate SCDs and identify high-risk groups viz. immediate family members prone/predisposed to SCAs/SCDs. This not only allows the high risk groups to take preventive measures but also informs them to take care of underlying modifiable risk factors. In essence, determining the cause of death in SCD cases is a vital step in caring for surviving relatives. A diagnosis of genetic cause can provide critical guidance in family screening and help prevent further SCA/SCD events.
In Survivors of sudden cardiac arrest (secondary prevention), or patients presenting with haemodynamically significant or sustained ventricular tachycardia (VT), detailed clinical assessment should be done to determine the underlying pathology. This includes a comprehensive history, 12 lead electrocardiogram, transthoracic echocardiogram, coronary imaging (eg. CT Angio, Cardiac Magnetic Resonance(CMR) etc), drug testing (e.g. sodium channel blocker for Brugada syndrome), stress test and genetic testing. Confirming a diagnosis allows management of the underlying disease and family screening if inherited heart disease is found. A secondary prevention ICD is recommended if no reversible pathology is found and there is no contraindication.
In those at risk of SCD (primary prevention) but who have not had an event, a diagnosis and disease‐specific therapy is used to manage any symptoms and can effectively reduce mortality. The risk stratification process is disease dependent and can include history (eg. family history of SCD), electrocardiographic features, LVEF, CMR imaging & electrophysiological studies.
Following a Sudden Unexplained Death, as a consequence of a hereditary pathology, family screening of first degree relatives should be performed. In cases where a cause has been identified in the decedent, a pathogenic genetic mutation may be identified in the proband, which may be used in cascade testing of first degree relatives.
Q24) What is an I.C.D.?
An implantable cardioverter defibrillator(ICD) also known as AICD (Automated Implantable Cardioverter Defibrillator) is a device implanted inside the body wherein it is able to perform the functions of cardioversion, defibrillation, and pacing of the heart when deemed necessary upon detection. It is therefore capable of correcting most life-threatening cardiac arrhythmias and has become the first-line treatment and prophylactic tool for patients who are at risk of sudden cardiac death due to potentially fatal abnormal heart rhythms like ventricular fibrillation and ventricular tachycardia.
Q25) How & where did its development come about?
The development of the ICD was pioneered at Sinai Hospital in Baltimore, USA by a team including Michel Mirowski, Morton Mower, Alois Langer and William Staewen. Mirowski teamed up with Mower and Staewen and together they commenced their research in 1969 but it was 11 years before they treated their first patient.
There was doubt that their ideas would ever become a clinical reality and work was commenced against much scepticism even by leading experts in the field of arrhythmias and sudden death citing that the implanted defibrillator system represents an imperfect solution in search of a plausible and practical application.
Q26) What were the challenges which the innovators faced during the developmental stages of ICD?
The problems to be overcome were the design of a system which would allow detection of ventricular fibrillation or ventricular tachycardia. Despite the lack of financial backing, they persisted and the first device was implanted in February 1980 at Johns Hopkins Hospital by Dr. Levi Watkins Jr.
The first devices required the chest to be cut open and a mesh electrode sewn onto the heart; pulse generator was placed in the abdomen.
Modern-day devices have been advanced to the extent that they detect abnormal heart rhythms and deliver therapy via programmable anti-tachycardia pacing in addition to low-energy and high-energy shocks.
Q27) What are the components of an I.C.D.?
ICDs are composed of an ICD generator and of wires. The first component or generator contains a computer chip or circuitry with RAM (memory), programmable software, a capacitor and a battery; this is implanted typically under the skin in the left upper chest.
The second part of the system is an electrode wire or wires that, similar to pacemakers, are connected to the generator and passed through a vein to the right chambers of the heart. The lead usually lodges in the apex or septum of the right ventricle. Just like pacemakers, ICDs can have a single wire or lead in the heart (in the right ventricle, single-chamber ICD), two leads (in the right atrium and right ventricle, dual-chamber ICD) or three leads (biventricular ICD, one in the right atrium, one in the right ventricle and one on the outer wall of the left ventricle).
Q28) What is the difference in their primary functional modality from that of a paceamaker?
The difference between pacemakers and ICDs is that pacemakers are also available as temporary units and are generally designed to correct slow heart rates, i.e.
bradycardia, while ICDs are often permanent safeguards against sudden life- threatening arrhythmias.
Q29) How does an I.C.D. look on a X-ray and how do we differentiate it from a pacemaker?
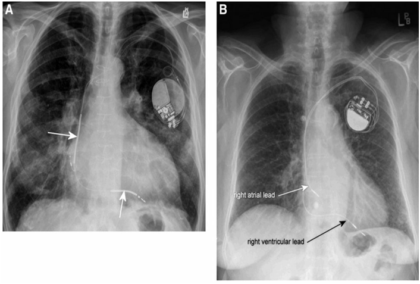
Shown above is the radiographic appearance of an implantable cardioverter-defibrillator i.e. I.C.D [A] and a cardiac pacemaker [B].
A reliable way to differentiate an ICD from a pacemaker is the presence of shocking coil(s) as seen indicated by the arrows in the right ventricle and sometimes in the Superior Vena Cava. These are seen as parts of the lead presenting as being more wider and radiodense.
Q30) What are the considerations to be kept in mind for ICD therapy?
ICDs carry the risk of acute procedural complications at implantation, such as pneumothorax, bleeding, acute lead dislodgement and cardiac tamponade as well as chronic risks, including infection, lead-related issues and inappropriate shocks.
For these reasons, balancing the benefits versus harms is important and should be discussed with the patient. Accurate risk stratification is critical to ensure adequate primary prevention without exposing people at low risk.
Q31) What are the indications for I.C.D. implantation?
Implantation of ICD is meant to prevent sudden cardiac death and is indicated in numerous conditions. These conditions are classified as primary and secondary prevention.
Those patients who have not suffered an arrhythmia episode which was life threatening in nature fall under Primary Prevention whereas Secondary prevention provides the strongest evidence for benefit and it is primarily for survivors of cardiac arrest secondary to ventricular fibrillation or hemodynamically unstable sustained ventricular tachycardia only after the reversible causes are excluded of course.
Q32) What purposes are served with an ICD implant?
Two broad but distinct classifications of conditions exist for whom ICD therapy is used – they fall into Primary Prevention & Secondary Prevention groups.
- Some conditions belonging to Primary prevention include prior myocardial infarction (within a minimum of 40 days ago) and LVEF lesser than or equal to 35%, Non-Ischaemic Dilated Cardiomyopathy with LVEF lesser than or equal to 35%, Long QT, Torsades on treatment, Brugada etc.
- Secondary Prevention entails conditions like prior episode of resuscitated VT/Vf or sustained hemodynamically unstable VT, episodes of spontaneous sustained ventricular tachycardia in conjunction with prevalent heart disease (valvular, iscahemic, hyperthrophic, dilated etc) and channelopathies.
Q33) What are the contraindications of ICD implantation?
ICDs are contraindicated in patients experiencing tachyarrhythmias with transient or reversible causes including but not limited to acute myocardial infarction, drug intoxication, drowning, electric shock, hypoxia, patients with incessant ventricular tachycardia or ventricular fibrillation and patients whose primary disorder is chronic atrial tachyarrhythmias with no concomitant VT or VF.
Q34) A conundrum - Is ICD therapy superior to treatment with Anti Arrhythmic drugs for managing and preventing sudden cardiac death?
This conundrum has however been laid to rest after numerous clinical trials & studies have demonstrated the superiority of the ICD by far over AAD (antiarrhythmic drugs) as a treatment modality altogether.
The SCD-HeFT trial (published in 2005) showed a significant all-cause mortality benefit for patients with ICD. Congestive heart failure patients that were implanted with an ICD had an all-cause death risk 23% lower than placebo and an absolute decrease in mortality of 7.2 percentage points after five
years in the overall population.
Reporting in 1999, the Antiarrhythmics Versus Implantable Defibrillators (AVID) trial consisted of 1,016 patients, and deaths in those treated with Anti Arrhythmic drugs were more frequent (n = 122) compared with deaths in the ICD groups (n = 80, p< 0.001).
In 2002 the MADITII trial showed benefit of ICD treatment in patients after myocardial infarction with reduced left ventricular function (EF<30).
Q35) What is MICRA?
Developed by Medtronic, it is the world’s smallest Transcatheter Pacing System approved for use in the United States by the FDA in April 2016 weighing 1.75grams, completely self contained within the heart, no leads required powered by an ultra-low circuit designed to deliver a 12 year battery life. It has a 99% plus implant success rate with 63% fewer complications.

Q36) What are the complications of associated with ICD implantation which have been drastically reduced/eliminated by MICRA?
Eliminated Pocket-related Complications:
- Infection
- Hematoma
Erosion Eliminated Lead-related Complications:
- Fractures
- Insulation breaches
- Venous thrombosis and obstruction
- Tricuspid regurgitation
Long-term Lead- and Pocket-related Complications with Traditional Systems:
- Pocket-related complications 8% at five years
- Lead-related complications 11% at five years
Although the leadless pacemaker is a relatively new technology, the preliminary reports of long-term performance and complications are promising, including low complications, few system revisions, and stable pacing parameters. These exciting new devices are not for all patients, however, and are contraindicated in those who require dual-chamber pacing, who have existing cardiovascular implantable electronic devices, mechanical tricuspid valves, pulmonary hypertension, or inferior vena cava (IVC) filters.
Q37) Is MICRA like devices a gamechanger of sorts in future?
While the future appears to be bright for these tiny-like devices, there are some concerns, including chronic right ventricular pacing due to single-chamber pacing potentially leading to atrioventricular and mechanical dysfunction and heart failure. Other noted drawbacks include the need for larger venous access tools for delivery, limited safety data from nonrandomized studies, and estimated but unproven longevity of the device.
Leadless pacing offers an innovative and safe alternative for cardiac pacing in patients who require single-chamber pacing while avoiding the common pitfalls of traditional transvenous pacemaker systems. Future avenues of development in this specialized area are boundless and include transforming kinetic energy from cardiac motion to fuel the pacemaker, wireless left ventricular pacing system, potentially utilizing leadless pacemakers with subcutaneous defibrillators, and the development of a leadless pacemaker with dual-chamber pacing capabilities and resynchronization therapy.
Q38) What can we infer and take back?
Sudden Cardiac/Sudden Cardiac Death is a major public health issue and among the top contributors to the global premature death burden. Although the incidence has declined with the advances in IHD management. Lesser gains have been made in the younger cohort and those with known cardiac disease and families of SCD victims. For families of SCD victims, a definitive diagnosis is paramount, where ever possible, genetic screening.
At a population level, By-stander BLS(basic life support)training and AED availability (Automatic External cardiac Defibrillator) have improved survivals rate in the western hemisphere, but further gains can be made globally with regular, high‐impact public health campaigns.
In essence, with USD 95 billion spent on medical research annually. In developing countries like India; It is imperative to acknowledge that a cumulative effort in terms of BLS/ACLS/CPR awareness, widespread availability of AEDs (Automatic External cardiac Defibrillator) and adoption of standardized protocols itself can certainly bring about a positive impact towards a rise in survival rates of Sudden Cardiac Arrests.
Dr Srikant Sharma, Senior Consultant Physician, and Dr Faraz Nawaz Resident Emergency; Moolchand Medcity, New Delhi

Disclaimer: This site is primarily intended for healthcare professionals. Any content/information on this website does not replace the advice of medical and/or health professionals and should not be construed as medical/diagnostic advice/endorsement or prescription. Use of this site is subject to our terms of use, privacy policy, advertisement policy. © 2020 Minerva Medical Treatment Pvt Ltd